Advertisements
Advertisements
प्रश्न
- Of the two gases, ammonia and hydrogen chloride, which is more dense? Name the method of collection of this gas.
- Give one example of a reaction between the above two gases which produce a solid compound.
- Write the balanced equation for a reaction in which ammonia is oxidized by:
- A metal oxide
- a gas which is not oxygen
उत्तर
- Hydrogen chloride is collected by the upward displacement of air.
- \[\ce{NH3 + HCl -> \underset{Ammonium chloride}{NH4Cl_{(s)}}}\]
-
- \[\ce{3CuO + 2NH3 ->[\Delta] 3Cu + N2 + 3H2O}\]
- \[\ce{8NH3 + 3Cl2 -> N2 + 6NH4Cl}\]
or \[\ce{2NH3 + 3Cl -> N2 + 6HCl}\]
APPEARS IN
संबंधित प्रश्न
What property of ammonia is demonstrated by the fountain experiment?
Give two reactions to show reducing property of ammonia.
Name a metallic chloride soluble in ammonium hydroxide.
How will you demonstrate the solubility of ammonia in water? Explain.
State your observation for the following cases
(i) Ammonia gas is burnt in an atomosphere of oxygen in the absence of a catalyst
(ii) Glass rod dipped in ammonium hydroxide is brought near the mouth of the concentrated hydrochloric acid bottle
The diagram shows a simple arrangement of the fountain experiment:
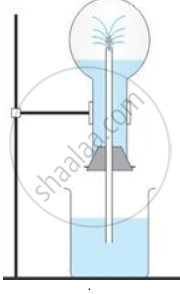
(i) Name the two gases you have studied which can be used in this experiment
(ii) What is the common properly demonstrated by this experiment
Choose the correct answer from the options given below :
Ammonia is produced when ammonium chloride is heated with
Write an equation to illustrate the reducing nature of ammonia.
State a relevant reason for the following:
Ammonia gas is not collected over water.
