Advertisements
Advertisements
प्रश्न
A steady current of 2 amperes was passed through two electrolytic cells X and Y connected in series containing electrolytes FeSO4and ZnSO4 until 2.8g of Fe deposited at the cathode of cell X. How long did the current flow? Calculate the mass of Zn deposited at the cathode of cell Y.
(Molar mass: Fe=56g mol-1,Zn=65.3g mol-1,1F=96500C mol-1)
उत्तर
I=2 A
W1= 2.8 g
Fe2+(aq)+2e-→Fe(s)
96500×2 C of charge is required to deposit=56 g of Fe
9650 C of charge is required to deposit=2.8 g of Fe
∵ Q `=1"t" "or" "t" 9650/2=4825"s"`
Using Faraday's second law of electrolysis,
`("W"_1("Weight of Fe deposited"))/("W"_2("Weight of Zn deposited")`
`=("E"_1("Equivalent weight of Fe"))/("E"_2("Equivalent weight of Fe")`
`2.8/"W"_2 =(56/5)/((65.3)/2)=56/65.3`
Or W2 = 3.265 g
APPEARS IN
संबंधित प्रश्न
State Kohlrausch’s law of independent migration of ions.
Why does the conductivity of a solution decrease with dilution?
The conductivity of sodium chloride at 298 K has been determined at different concentrations and the results are given below:
| Concentration/M | 0.001 | 0.010 | 0.020 | 0.050 | 0.100 |
| 102 × κ/S m−1 | 1.237 | 11.85 | 23.15 | 55.53 | 106.74 |
Calculate `∧_"m"`for all concentrations and draw a plot between `∧_"m"`and `"c"^(1/2)`. Find the value of `∧_"m"^0`.
Kohlrausch law of independent migration of ions states ____________.
Assertion: `"E"_("Ag"^+ //"Ag")` increases with increase in concentration of Ag+ ions.
Reason: `"E"_("Ag"^+ //"Ag")` has a positive value.
The molar conductance of \[\ce{NaCl, HCl}\] and \[\ce{CH3COONa}\] at infinite dilution are 126.45, 426.16 and 91.0 S cm2 mol−1 respectively. The molar conductance of \[\ce{CH3COOH}\] at infinite dilution is. Choose the right option for your answer.
The molar conductivity of CH3COOH at infinite dilution is 390 Scm2/mol. Using the graph and given information, the molar conductivity of CH3COOK will be:
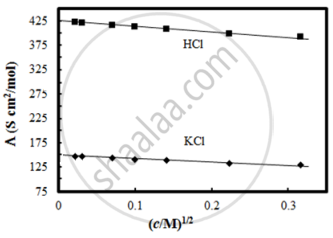
Molar conductivity of substance “A” is 5.9 × 103 S/m and “B” is 1 × 10–16 S/m. Which of the two is most likely to be copper metal and why?
Assertion (A) : Conductivity decreases with decrease in concentration of electrolyte.
Reason (R) : Number of ions per unit volume that carry the current in a solution decreases on dilution.
The resistance of a conductivity cell with a 0.1 M KCl solution is 200 ohm. When the same cell is filled with a 0.02 M NaCl solution, the resistance is 1100 ohm. If the conductivity of 0.1 M KCl solution is 0.0129 ohm-1 cm-1, calculate the cell constant and molar conductivity of 0.02 M NaCl solution.
