Advertisements
Advertisements
प्रश्न
The conductivity of sodium chloride at 298 K has been determined at different concentrations and the results are given below:
| Concentration/M | 0.001 | 0.010 | 0.020 | 0.050 | 0.100 |
| 102 × κ/S m−1 | 1.237 | 11.85 | 23.15 | 55.53 | 106.74 |
Calculate `∧_"m"`for all concentrations and draw a plot between `∧_"m"`and `"c"^(1/2)`. Find the value of `∧_"m"^0`.
उत्तर
| Concentration (M) | C1/2 (M1/2) |
κ(S m−1) | κ(S cm−1) | `∧_"m" = (κ xx 1000)/"M"` (S cm2 mol−1) |
| 0.001 | 0.0316 | 1.237 × 10−2 | 1.237 × 10−4 | `∧_"m" = (1.237 xx 10^-4 xx 1000)/0.001` = 123.7 |
| 0.010 | 0.100 | 11.85 × 10−2 | 11.85 × 10−4 | `∧_"m" = (11.85 xx 10^-4 xx 1000)/(0.010)` = 118.5 |
| 0.020 | 0.141 | 23.15 × 10−2 | 23.15 × 10−4 | `∧_"m" = (23.15 xx 10^-4 xx 1000)/0.020` = 115.8 |
| 0.050 | 0.224 | 55.53 × 10−2 | 55.53 × 10−4 | `∧_"m" = (55.53 xx 10^-4 xx 1000)/(0.1050)` = 111.1 |
| 0.100 | 0.316 | 106.74 × 10−2 | 106.74 × 10−4 | `∧_"m" = (106.74 xx 10^-4 xx 1000)/(0.100)` = 106.7 |
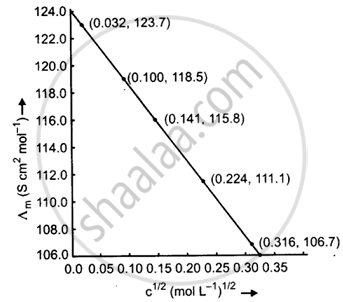
When the straight line is drawn backwards, it meets the `∧_"m"`axis at 124.0 S cm2 mol−1, which is the value of `∧_"m"^0`.
APPEARS IN
संबंधित प्रश्न
Define "Molar conductivity".
Why conductivity of an electrolyte solution decreases with the decrease in concentration ?
The S.I. unit of cell constant for conductivity cell is __________.
In the plot of molar conductivity (∧m) vs square root of concentration (c1/2) following curves are obtained for two electrolytes A and B : 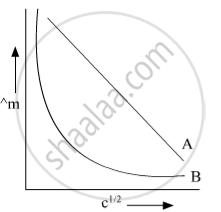
Answer the following:
(i) predict the nature of electrolytes A and B.
(ii) What happens on the extrapolation of ∧m to concentration approaching for electrolytes A and B?
Molar conductivity denoted by the symbol Λm is related to the conductivity of the solution by the equation (k is the conductivity and c is the concentration).
Conductivity always decreases with decrease in concentration both, for weak and strong electrolytes because of the fact that ____________.
Which of the statements about solutions of electrolytes is not correct?
Write the cell reaction of a lead storage battery when it is discharged. How does the density of the electrolyte change when the battery is discharged?
Assertion: Copper sulphate can be stored in zinc vessel.
Reason: Zinc is less reactive than copper.
Solutions of two electrolytes ‘A’ and ‘B’ are diluted. The Λm of ‘B’ increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Justify your answer. Graphically show the behavior of ‘A’ and ‘B’.
An increase in equivalent conductance of a strong electrolyte with dilution is mainly due to :-
Which of the following halogen acids is the strongest reducing agent?
Which of the following increases with the increase in the concentration of the solution?
The molar conductivity of CH3COOH at infinite dilution is 390 Scm2/mol. Using the graph and given information, the molar conductivity of CH3COOK will be:
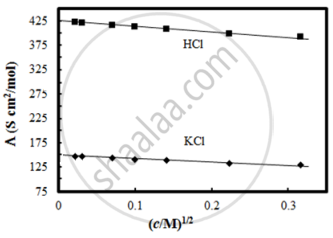
Given below are two statements:
Statements I: The limiting molar conductivity of KCl (strong electrolyte) is higher compared to that of CH3COOH (weak electrolyte).
Statement II: Molar conductivity decreases with decrease in concentration of electrolyte.
In the light of the above statements, choose the most appropriate answer from the options given below:
The specific conductance of 2.5 × 10-4 M formic acid is 5.25 × 10-5 ohm-1 cm-1. Calculate its molar conductivity and degree of dissociation.
Given `λ°_("H"^+)` = 349.5 ohm-1 cm2 mol-1 and
`λ°_("HCOO"^-) = 50.5 " ohm"^-1 "cm"^2 "mol"^-1`
The solution of two electrolytes A and B are diluted. ^m of B increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Give a reason.
