Advertisements
Advertisements
प्रश्न
Assertion: Copper sulphate can be stored in zinc vessel.
Reason: Zinc is less reactive than copper.
पर्याय
Both assertion and reason are true and the reason is the correct explanation of assertion.
Both assertion and reason are true and the reason is not the correct explanation of assertion.
Assertion is true but the reason is false.
Both assertion and reason are false.
Assertion is false but reason is true.
उत्तर
Both assertion and reason are false.
Explanation:
Zinc will get dissolved in CuS04 solution since zinc is more reactive than copper.
APPEARS IN
संबंधित प्रश्न
State Kohlrausch’s law of independent migration of ions.
State Kohlrausch Law
How can you determine limiting molar conductivity, 0 m for strong electrolyte and weak electrolyte?
In the plot of molar conductivity (∧m) vs square root of concentration (c1/2), following curves are obtained for two electrolytes A and B:
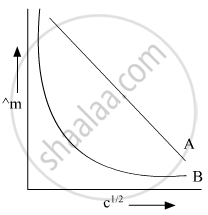
Answer the following:
(i) Predict the nature of electrolytes A and B.
(ii) What happens on extrapolation of ∧m to concentration approaching zero for electrolytes A and B?
When acidulated water (dil.H2SO4 solution) is electrolysed, will the pH of the solution be affected? Justify your answer.
Write the cell reaction of a lead storage battery when it is discharged. How does the density of the electrolyte change when the battery is discharged?
The limiting molar conductivities for Nacl, KBr and KCI are 126, 152 and 150 S cm2 mol–1 respectively. The limiting molar conductivity for Na Br is:-
Which of the following increases with the increase in the concentration of the solution?
Molar conductivity of substance “A” is 5.9 × 103 S/m and “B” is 1 × 10–16 S/m. Which of the two is most likely to be copper metal and why?
The solution of two electrolytes A and B are diluted. ^m of B increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Give a reason.
