Advertisements
Advertisements
प्रश्न
How can you determine limiting molar conductivity, 0 m for strong electrolyte and weak electrolyte?
उत्तर
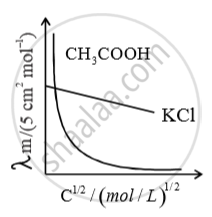
For strong electrolyte, plot of m
against c, extrapolated to the y-axis, gives the value of m for
strong electrolyte. For weak electrolyte, intercept is not obtained as o m
cannot be extra apolated to zero conc. hence limiting molar conductivity of weak electrolyte is obtained from that of strong electrolytes. Using Kohlrausch law of independent migration of ions.
`λ_m^0(CH_3COOH) = lamda^0CH_3COONa + lamda_{HCl}^circ - lamda_{NaCl}^circ`
`lamda_m^circ(CH_3COOH) = lamda_{CH_3COO-}^circ + lamda_{H^+}^circ`
APPEARS IN
संबंधित प्रश्न
Why does the conductivity of a solution decrease with dilution?
The molar conductivity of 0.025 mol L−1 methanoic acid is 46.1 S cm2 mol−1. Calculate its degree of dissociation and dissociation constant. Given \[\ce{λ^0_{(H^+)}}\] = 349.6 S cm2 mol−1 and \[\ce{λ^0_{(HCOO^-)}}\] = 54.6 S cm2 mol−1.
Conductivity of 0.00241 M acetic acid is 7.896 × 10−5 S cm−1. Calculate its molar conductivity and if `∧_"m"^0` for acetic acid is 390.5 S cm2 mol−1, what is its dissociation constant?
Write mathematical expression of molar conductivity of the given solution at infinite dilution.
Which of the statements about solutions of electrolytes is not correct?
\[\ce{Λ^0_m}_{(NH_4OH)}\] is equal to ______.
\[\ce{Λ^0_m H2O}\] is equal to:
(i) \[\ce{Λ^0_m_{(HCl)} + \ce{Λ^0_m_{(NaOH)} - \ce{Λ^0_m_{(NaCl)}}}}\]
(ii) \[\ce{Λ^0_m_{(HNO_3)} + \ce{Λ^0_m_{(NaNO_3)} - \ce{Λ^0_m_{(NaOH)}}}}\]
(iii) \[\ce{Λ^0_{(HNO_3)} + \ce{Λ^0_m_{(NaOH)} - \ce{Λ^0_m_{(NaNO_3)}}}}\]
(iv) \[\ce{Λ^0_m_{(NH_4OH)} + \ce{Λ^0_m_{(HCl)} - \ce{Λ^0_m_{(NH_4Cl)}}}}\]
Assertion: Λm for weak electrolytes shows a sharp increase when the electrolytic solution is diluted.
Reason: For weak electrolytes degree of dissociation increases with dilution of solution.
Which of the following halogen acids is the strongest reducing agent?
Suggest a way to determine the `∧_"m"^∘`value of water.
