Advertisements
Advertisements
प्रश्न
How can you determine limiting molar conductivity, 0 m for strong electrolyte and weak electrolyte?
उत्तर
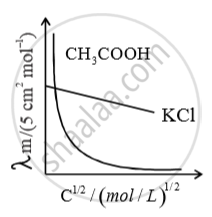
For strong electrolyte, plot of m
against c, extrapolated to the y-axis, gives the value of m for
strong electrolyte. For weak electrolyte, intercept is not obtained as o m
cannot be extra apolated to zero conc. hence limiting molar conductivity of weak electrolyte is obtained from that of strong electrolytes. Using Kohlrausch law of independent migration of ions.
`λ_m^0(CH_3COOH) = lamda^0CH_3COONa + lamda_{HCl}^circ - lamda_{NaCl}^circ`
`lamda_m^circ(CH_3COOH) = lamda_{CH_3COO-}^circ + lamda_{H^+}^circ`
APPEARS IN
संबंधित प्रश्न
Define "Molar conductivity".
The conductivity of 0.20 M solution of KCl at 298 K is 0.025 S cm−1. Calculate its molar conductivity.
Write mathematical expression of molar conductivity of the given solution at infinite dilution.
In the plot of molar conductivity (∧m) vs square root of concentration (c1/2), following curves are obtained for two electrolytes A and B:
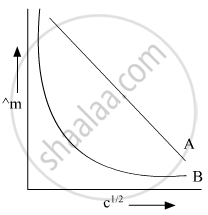
Answer the following:
(i) Predict the nature of electrolytes A and B.
(ii) What happens on extrapolation of ∧m to concentration approaching zero for electrolytes A and B?
Molar conductivity denoted by the symbol Λm is related to the conductivity of the solution by the equation (k is the conductivity and c is the concentration).
Solutions of two electrolytes ‘A’ and ‘B’ are diluted. The Λm of ‘B’ increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Justify your answer.
Assertion (A): Molar conductivity decreases with increase in concentration.
Reason (R): When concentration approaches zero, the molar conductivity is known as limiting molar conductivity.
The following questions are case-based questions. Read the passage carefully and answer the questions that follow:
| Rahul set up an experiment to find the resistance of aqueous KCl solution for different concentrations at 298 K using a conductivity cell connected to a Wheatstone bridge. He fed the Wheatstone bridge with a.c. power in the audio frequency range 550 to 5000 cycles per second. Once the resistance was calculated from the null point, he also calculated the conductivity K and molar conductivity ∧m and recorded his readings in tabular form. |
| S. No. | Conc. (M) |
k S cm−1 | ∧m S cm2 mol−1 |
| 1. | 1.00 | 111.3 × 10−3 | 111.3 |
| 2. | 0.10 | 12.9 × 10−3 | 129.0 |
| 3. | 0.01 | 1.41 × 10−3 | 141.0 |
Answer the following questions:
(a) Why does conductivity decrease with dilution? (1)
(b) If `∧_"m"^0` of KCl is 150.0 S cm2 mol−1, calculate the degree of dissociation of 0.01 M KCI. (1)
(c) If Rahul had used HCl instead of KCl then would you expect the ∧m values to be more or less than those per KCl for a given concentration? Justify. (2)
OR
(c) Amit a classmate of Rahul repeated the same experiment with CH3COOH solution instead of KCl solution. Give one point that would be similar and one that would be different in his observations as compared to Rahul. (2)
The resistance of a conductivity cell with a 0.1 M KCl solution is 200 ohm. When the same cell is filled with a 0.02 M NaCl solution, the resistance is 1100 ohm. If the conductivity of 0.1 M KCl solution is 0.0129 ohm-1 cm-1, calculate the cell constant and molar conductivity of 0.02 M NaCl solution.
The solution of two electrolytes A and B are diluted. ^m of B increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Give a reason.
