Advertisements
Advertisements
प्रश्न
An aromatic compound ‘A’ on treatment with aqueous ammonia and heating forms compound ‘B’ which on heating with Br2 and KOH forms a compound ‘C’ of molecular formula C6H7N. Write the structures and IUPAC names of compounds A, B and C.
उत्तर
It is given that compound ‘C’ having the molecular formula C6H7N is formed by heating compound ‘B’ with Br2 and KOH. This is a Hoffmann bromamide degradation reaction. Therefore, compound ‘B’ is an amide and compound ‘C’ is an amine. The only amine having the molecular formula C6H7N is aniline (C6H5NH2).
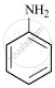
Therefore, compound ‘B’ (from which ‘C’ is formed) must be benzamide (C6H5CONH2).
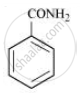
Further, benzamide is formed by heating compound ‘A’ with aqueous ammonia. Therefore, compound ‘A’ must be benzoic acid.
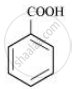
The given reactions can be explained with the help of the following equations:

APPEARS IN
संबंधित प्रश्न
Convert aniline into 1, 3, 5-tribromobenzene.
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
(CH3)2CHNH2
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
CH3NHCH(CH3)2
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
(CH3)3CNH2
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
(CH3CH2)2NCH3
Give one chemical test to distinguish between the following pair of compounds.
Secondary and tertiary amines
Give one chemical test to distinguish between the following pair of compounds.
Ethylamine and aniline
Account for the following:
pKb of aniline is more than that of methylamine.
Account for the following:
Methylamine in water reacts with ferric chloride to precipitate hydrated ferric oxide.
How will you convert methanol to ethanoic acid?
How will you convert methanamine into ethanamine?
Accomplish the following conversion:
Benzoic acid to aniline
Complete the following reactions:
Give reasons Although –NH2 is o/p directing group, yet aniline on nitration gives a significant amount of m-nitroaniline
Do the following conversions in not more than two steps :
Propanone to Propene
Give simple chemical tests to distinguish between the following pair of compounds:
Benzaldehyde and Benzoic acid
Do the following conversions in not more than two steps:

Predict the product 'A' in the following reaction.
\[\ce{\underset{(Acetyl chloride)}{CH3COCl}->[H2][Pd-BaSO4]A + HCl}\]
