Advertisements
Advertisements
प्रश्न
An element X has a valency of 2. Write the simplest formula for oxide of the element.
उत्तर
Valency of X = 2
Valency of Oxygen = 2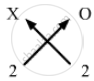
Formula for oxide of element X can be worked out by crossing the valencies of the constituent elements.
Therefore, formula for oxide of X is XO.
APPEARS IN
संबंधित प्रश्न
Calculate the number of aluminium ions present in 0.051 g of aluminium oxide.
(Hint: The mass of an ion is the same as that of an atom of the same element. Atomic mass of Al = 27 u)
Name the following radicals :
OH
Write the names of `"ZnCl"_2 ` compounds :
Fill in the following blank with suitable word:
The particle which is formed by the loss of electrons by an atom is called..............
Give example.
Basic radicals
What do you understand by the following terms?
Basic radical
Write the basic radicals and acidic radicals of the following and then write the chemical formulae of these compound.
Magnesium phosphate
Differentiate between the following with suitable examples- A basic radical and an acid radical
Select the correct answer to the statement given below:
An elementary particle of matter – of a group of atoms of different elements behaving as a single unit with charge on the group.
Name the following:
The radical containing one nitrogen and four hydrogen atoms.
