Advertisements
Advertisements
प्रश्न
An element X has a valency of 2. Write the simplest formula for oxide of the element.
उत्तर
Valency of X = 2
Valency of Oxygen = 2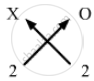
Formula for oxide of element X can be worked out by crossing the valencies of the constituent elements.
Therefore, formula for oxide of X is XO.
APPEARS IN
संबंधित प्रश्न
Name the following radicals :
HC03
The valency of calcium is 2. Write the valencies of other radical in the following :
- CaO
- Ca(OH)2
- CaC03
- CaCl2
A liquid compound X of molecular mass 18 u can be obtained from a number of natural sources. All the animals and plants need liquid X for their survival. When an electric current is passed through 200 grams of pure liquid X under suitable conditions, then 178 grams of gas Y and 22 grams of gas Z are produced. Gas Y is produced at the positive electrode whereas gas Z is obtained at the negative electrode. Moreover, gas Y supports combustion whereas gas Z burns itself causing explosions.
- Name (i) liquid X (ii) gas Y, and (iii) gas Z.
- What is the ratio of the mass of element Z to the mass of element Y in the liquid X ?
- Which law of chemical combination is illustrated by this example ?
- Name two sources of liquid X.
- State an important use of Y in our life.
What is an ion ? How is an ion formed ? Explain with the help of two examples of different ions.
What do you understand by the following terms?
Acid radical
What do you understand by the following terms?
Basic radical
Name the following:
The radical containing one nitrogen and four hydrogen atoms.
State if the following element or radical is divalent?
Bisulphite
Match the names of ions and radicals from 1 to 10 with their correct answer from A to Q.
| Column A | Column B |
| 1. Hypochlorite | A: Hg2+ |
| 2. Permanganate | B: \[\ce{MnO_4^1-}\] |
| 3. Plumbous | C: Sn4+ |
| 4. Zincate | D: Pb2+ |
| 5. Nitride | E: Sn2+ |
| 6. Mercuric | F: Pb4+ |
| 7. Stannic | G: \[\ce{SO_3^2-}\] |
| 8. Nitrite | H: N3- |
| 9. Sulphite | I: \[\ce{NO_2^1-}\] |
| 10. Dichromate | J: \[\ce{MnO_4^2-}\] |
| K: Hg1+ | |
| L: \[\ce{SO_4^2-}\] | |
| M: CIO1- | |
| N: \[\ce{ZnO_2^2-}\] | |
| O: \[\ce{Cr_2O_7^2-}\] | |
| P: \[\ce{CrO_4^2-}\] | |
| Q: \[\ce{ClO_3^1-}\] |
Atoms that carry positive or negative charges are called ______.
