Advertisements
Advertisements
प्रश्न
An element X has a valency of 2. Write the simplest formula for bromide of the element.
उत्तर
Valency of X = 2
Valency of Br = 1
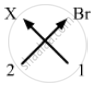
Formula for bromide of element X can be worked out by crossing the valencies of the constituent elements.
Therefore, formula for bromide of X is: XBr2.
APPEARS IN
संबंधित प्रश्न
What are polyatomic ions? Give examples.
Write the names of `"HgO"` compounds :
State whether the following statements are true or false:
A sodium ion has positive charge because it has more protons than a neutral atom.
State whether the following statements are true or false :
A chloride ion has negative charge because it has more electrons than a neutral atom.
What is an ion ? How is an ion formed ? Explain with the help of two examples of different ions.
Write the basic radicals and acidic radicals of the following and then write the chemical formulae of these compounds.
Barium sulphate
Write the basic radicals and acidic radicals of the following and then write the chemical formulae of these compound.
Stannic phosphate
State if the following element or radical is divalent?
Ammonium
State if the following element or radical is divalent?
Carbide
State if the following element or radical is divalent?
Bisulphite
