Advertisements
Advertisements
Question
An element X has a valency of 2. Write the simplest formula for bromide of the element.
Solution
Valency of X = 2
Valency of Br = 1
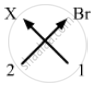
Formula for bromide of element X can be worked out by crossing the valencies of the constituent elements.
Therefore, formula for bromide of X is: XBr2.
APPEARS IN
RELATED QUESTIONS
Write the names of `"NH"_4"OH"` compounds :
Write the names of `"H"_2"S"` compounds :
Fill in the following blank with suitable word:
A sulphide ion has negative charge because it contains less ....................... than ...................
The valencies (or charges) of some of the ions are given below :
Ion Valency Ion Valency (Charge) (Charge)
Sodium ion 1+ Nitrate ion 1–
Copper ion 2+ Sulphide ion 2–
Using this information, write down the formulae of :
- Sodium sulphide
- Copper nitrate
Write symbols of the following elements and the radicals obtained from them and indicate the charge on the radicals.
Mercury, potassium, nitrogen, copper, sulphur, carbon, chlorine, oxygen
Write the basic radicals and acidic radicals of the following and then write the chemical formulae of these compound.
Ferrous sulphide
Write the basic radicals and acidic radicals of the following and then write the chemical formulae of these compound.
Stannic oxide
Match the radicals in List I with their correct names from List II.
| List I | List II | |
| 1. `"NO"_3^(1-)` | A: Bisulphite | F: Sulphate |
| 2. `"HSO"_4^(1-)` | B: Phosphate | G: Nitrate |
| 3. `"SO"_4^(2-)` | C: Carbonate | H: Phosphite |
| 4. `"PO"_4^(3-)` | D: Nitrite | I: Sulpite |
| 5. `"HCO"_3^(1-)` | E: Bicarbonate | J: Bisulphate |
State if the following element or radical is divalent?
Chromium
State if the following element or radical is divalent?
Permanganate
