Advertisements
Advertisements
प्रश्न
An element X of group 15 exists as diatomic molecule and combines with hydrogen at 773 K in presence of the catalyst to form a compound, ammonia which has a characteristic pungent smell.
- Identify the element X. How many valence electrons does it have?
- Draw the electron dot structure of the diatomic molecule of X. What type of bond is formed in it?
- Draw the electron dot structure for ammonia and what type of bond is formed in it?
उत्तर
- Element X is nitrogen. Nitrogen has 5 valence electrons.
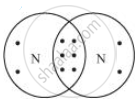
Nitrogen molecule has covalent bonds in between the nitrogen atoms.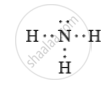
Ammonia molecule ahs covalent bonds between the nitrogen and hydrogen atoms.
APPEARS IN
संबंधित प्रश्न
Rewrite the following statement after correction, if necessary:
Groups have elements with consecutive atomic numbers.
An element X is in group 2 of the periodic table what will be the formula of its oxide?
An atom has the electron structure of 2, 7.
(a) What is the atomic number of this atom?
(b) To which of the following would it be chemically similar?
7N, 15P, 17Cl, 18Ar
(c) Why would you expect it to be similar?
An element X from group 2 reacts with element Y from group 16 of the periodic table.
(a) What is the formula of the compound formed?
(b) What is the nature of bond in the compound formed?
Find the neutral atom in the periodic table which has the same number of electrons as K+ and Cl−. What is this number?
An element X from group 2 of the Periodic Table reacts with Y from group 17 to form a compound. Give the formula of the compound.
Which of the following property will be common in group 1 elements?
Which type of elements are poor conductors of heat and electricity?
An element is placed in 2nd Group and 3rd Period of the Periodic Table, burns in presence of oxygen to form a basic oxide.
- Identify the element
- Write the electronic configuration
- Write the balanced equation when it burns in the presence of air
- Write a balanced equation when this oxide is dissolved in water
- Draw the electron dot structure for the formation of this oxide
Relate the names of the following scientists with the statements given below.
Arranged elements into groups containing three elements each.
