Advertisements
Advertisements
प्रश्न
An element X of group 15 exists as diatomic molecule and combines with hydrogen at 773 K in presence of the catalyst to form a compound, ammonia which has a characteristic pungent smell.
- Identify the element X. How many valence electrons does it have?
- Draw the electron dot structure of the diatomic molecule of X. What type of bond is formed in it?
- Draw the electron dot structure for ammonia and what type of bond is formed in it?
उत्तर
- Element X is nitrogen. Nitrogen has 5 valence electrons.
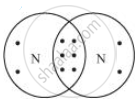
Nitrogen molecule has covalent bonds in between the nitrogen atoms.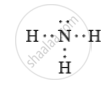
Ammonia molecule ahs covalent bonds between the nitrogen and hydrogen atoms.
APPEARS IN
संबंधित प्रश्न
State whether the following statement is true of false:
The elements in a group have consecutive atomic numbers.
An element X is in group 2 of the periodic table What will be the formula of its chloride?
As element X has mass number 40 and contains 21 neutrons in its atom. To which group of the periodic table does it belong?
The elements A, B and C belong to groups 1, 14, and 17 respectively of the periodic table.
(a) Which two elements will form a covalent compound?
(b) Which two elements will form an ionic compound?
Match the following
| 1. | Galvanisation | Noble gas elements |
| 2. | Calcination | Coating with Zn |
| 3. | Redox reaction | Silver-tin amalgam |
| 4. | Dental filling | Alumino thermic process |
| 5. | Group 18 elements | Heating in the absence of air |
Which one of the following statements is not correct about the trends in the properties of the elements of a period on going from left to right?
The elements A, B, and C belong to groups 1, 14, and 17 respectively of the Periodic Table. Which two elements will form ionic compounds?
Which of the following elements does not lose an electron easily?
Which type of elements are poor conductors of heat and electricity?
Identify the following:
An element in Period 1 which can be placed in both Group 1 and Group 17 of the Periodic Table.
