Advertisements
Advertisements
Question
An element X of group 15 exists as diatomic molecule and combines with hydrogen at 773 K in presence of the catalyst to form a compound, ammonia which has a characteristic pungent smell.
- Identify the element X. How many valence electrons does it have?
- Draw the electron dot structure of the diatomic molecule of X. What type of bond is formed in it?
- Draw the electron dot structure for ammonia and what type of bond is formed in it?
Solution
- Element X is nitrogen. Nitrogen has 5 valence electrons.
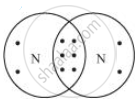
Nitrogen molecule has covalent bonds in between the nitrogen atoms.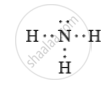
Ammonia molecule ahs covalent bonds between the nitrogen and hydrogen atoms.
APPEARS IN
RELATED QUESTIONS
Rewrite the following statement after correction, if necessary:
Isotopes are the elements of the same group.
Why do we classify elements?
Write the names and symbols of two very reactive metals belonging to group 1 of the periodic table. Explain by drawing electronic structure, how either one of the two metals reacts with a halogen. With which name is the bond formed between these elements known and what is the class of the compound so formed known? State any four physical properties of such compounds.
The molecular formula of the chloride of element X is XCl. This compound is a solid having a high melting point. Which of the following elements be present in the same group as X.
In which block of the modern periodic table are the nonmetals found?
Write the name.
The atom having smallest atomic radius from group 1.
Example for liquid metal is ______.
Which one of the following statements is not correct about the trends in the properties of the elements of a period on going from left to right?
An element with the atomic number 40 is placed in the period.
Which type of elements are poor conductors of heat and electricity?
