Advertisements
Advertisements
प्रश्न
An ether (A), C5H12O, when heated with excess of hot HI produce two alkyl halides which on hydrolysis form compound (B) and (C), oxidation of (B) gave and acid (D), whereas oxidation of (C) gave a ketone (E). Deduce the structural formula of (A), (B), (C), (D), and (E).
उत्तर
i. The ether (A) with molecular formula C5H12O is
\[\begin{array}{cc}\ce{H3C - CH2 - O - HC -CH3}\\\phantom{............}|\phantom{}\\ \phantom{............}\ce{CH3\phantom{}}\end{array}\]
ii. Reacts with hot HI to produce two alkyl halides as follows:
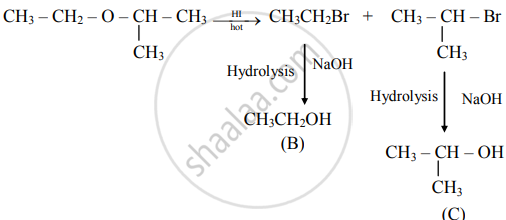
iii. Oxidation of (B) gives acid
\[\ce{CH3 - CH2 - OH ->[{[O]}] \underset{\text{(D)}}{CH3COOH}}\]
iv. Oxidation of (C) gives ketone
\[\begin{array}{cc}
\ce{CH3 - CH - OH ->[{[O]}] CH3 - C = O}\\
\phantom{...}|\phantom{....................}|\\
\phantom{....}\ce{CH3}\phantom{...........}\ce{(E)}\phantom{...}\ce{CH3}
\end{array}\]
Hence, structural formulae of compounds (A) to (E) are
(A)
\[\begin{array}{cc}\ce{CH3 - CH2 - O - CH - CH3} \\\phantom{.......................}|\phantom{...........}\\ \phantom{.......................}\ce{CH3\phantom{...........}}\end{array}\]
(2-Ethoxypropane)
(B)
CH3CH2–OH (Ethanol)
(c)
\[\begin{array}{cc}\ce{CH3 - CH - CH3}\\|\phantom{}\\ \ce{OH\phantom{}}\end{array}\]
(Propan-2-ol)
(D)
CH3COOH (Ethanoic acid)
(E)
\[\begin{array}{cc}
\ce{CH3 - CH - CH3}\\
\phantom{...}||\phantom{.....}\\
\phantom{...}
\ce{O}\phantom{.....}\end{array}\]
(Propanone)
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Which is the most resistant alcohol towards oxidation reaction among the following?
The reagents used to convert phenol to 2,4,6-tribromophenol is _____________
Write IUPAC name of crotonyl alcohol.
Write Kolbe’s reaction.
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
Which alcohol is difficult to oxidise?
Which of the following compounds is obtained, when phenol react with bromine water?
In phenols, −OH group is attached to ___________ hybridised carbon.
Bromination of phenol, will NOT give:
Phenol is ____________.
Isobutylene on hydroboration followed by oxidation with hydrogen peroxide in presence of base yields ______.
Cumene is used in the commercial method for the manufacture of ____________.
What is the product of the following reaction?
\[\ce{CH3 - CH2 - CH2 - OH ->[conc. H2SO4][\Delta]}\]
\[\ce{CH3 - CH = CH2 ->[HBr][Peroxide] A}\]
Identify A.
Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives __________.
Which of the following reagents is used to convert phenol to benzene?
What is INCORRECT about the product written in the below given reaction?
\[\ce{R - CH2 - OH ->[PCC] R - COOH}\]
Which of the following conversion explains the acidic nature of alcohols?
The number of σ bonds in carbolic acid is ______.
Identify the reagent A used in the following conversion.
\[\ce{Phenol ->[A] Benzoquinone}\]
Which of the following compounds has lowest boiling point?
Which among the following is not the method of preparation of phenol?
What is the product obtained in Reimer - Tiemann reaction?
The product C in the following reaction is

The major product obtained in the following reaction is

Arrange O - H, C - H and N - H bonds in increasing order of their bond polarity.
Write the product when 1°, 2° and 3° alcohol vapours are passed over hot copper.
Write chemical reaction when hot copper is treated with Vapours of 3° (tertiary) alcohol.
