Advertisements
Advertisements
प्रश्न
An ether (A), C5H12O, when heated with excess of hot HI produce two alkyl halides which on hydrolysis form compound (B) and (C), oxidation of (B) gave and acid (D), whereas oxidation of (C) gave a ketone (E). Deduce the structural formula of (A), (B), (C), (D), and (E).
उत्तर
i. The ether (A) with molecular formula C5H12O is
\[\begin{array}{cc}\ce{H3C - CH2 - O - HC -CH3}\\\phantom{............}|\phantom{}\\ \phantom{............}\ce{CH3\phantom{}}\end{array}\]
ii. Reacts with hot HI to produce two alkyl halides as follows:
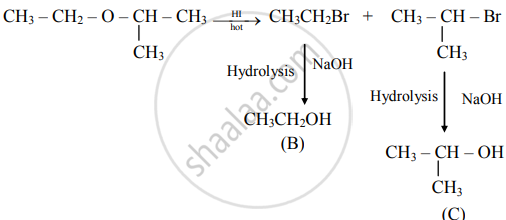
iii. Oxidation of (B) gives acid
\[\ce{CH3 - CH2 - OH ->[{[O]}] \underset{\text{(D)}}{CH3COOH}}\]
iv. Oxidation of (C) gives ketone
\[\begin{array}{cc}
\ce{CH3 - CH - OH ->[{[O]}] CH3 - C = O}\\
\phantom{...}|\phantom{....................}|\\
\phantom{....}\ce{CH3}\phantom{...........}\ce{(E)}\phantom{...}\ce{CH3}
\end{array}\]
Hence, structural formulae of compounds (A) to (E) are
(A)
\[\begin{array}{cc}\ce{CH3 - CH2 - O - CH - CH3} \\\phantom{.......................}|\phantom{...........}\\ \phantom{.......................}\ce{CH3\phantom{...........}}\end{array}\]
(2-Ethoxypropane)
(B)
CH3CH2–OH (Ethanol)
(c)
\[\begin{array}{cc}\ce{CH3 - CH - CH3}\\|\phantom{}\\ \ce{OH\phantom{}}\end{array}\]
(Propan-2-ol)
(D)
CH3COOH (Ethanoic acid)
(E)
\[\begin{array}{cc}
\ce{CH3 - CH - CH3}\\
\phantom{...}||\phantom{.....}\\
\phantom{...}
\ce{O}\phantom{.....}\end{array}\]
(Propanone)
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Which is the most resistant alcohol towards oxidation reaction among the following?
Write two points of difference between the properties of phenol and ethyl alcohol.
Oxidation of ethyl alcohol using K2Cr2O7/dil H2SO4 leads to formation of _______________
Arrange the following in decreasing order of acid strength.
CH3OH, CH3–CH2–OH, CH3–CH(OH)–CH3, (CH3)3–C–OH
Write IUPAC name of crotonyl alcohol.
What is the action of following reagents on pent-3-enal?
- H2/Ni
- LiAlH4/H3O+
Name the reagents used to convert phenol into
- picric acid
- p-benzoquinone
What is the action of conc. H2SO4 on carbolic acid at 373 K.
An organic compound gives hydrogen on reaction with sodium metal. It forms an aldehyde with molecular formula C2H4O on oxidation with pyridinium chlorochromate. Give the chemical equations in support of these observations.
Which alcohol is difficult to oxidise?
α-butylene when subjected to hydroboration oxidation reaction, yields ______.
Phenol is obtained from cumene ____________.
Sodium metal with ethyl alcohol gives __________ gas.
Bromination of phenol, will NOT give:
Which of following elements does not react with hot concentrated sulphuric acid?
What is the product of the following reaction?
\[\ce{CH3 - CH2 - CH2 - OH ->[conc. H2SO4][\Delta]}\]
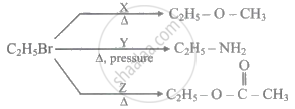
Identify reagents X, Y and Z.
Which of the following reagents is used to convert phenol to benzene?
The number of moles of hydrogen gas formed when 2 moles of 2-methylpropan-2-ol reacts with aluminium is ____________.
What is INCORRECT about the product written in the below given reaction?
\[\ce{R - CH2 - OH ->[PCC] R - COOH}\]
Identify the reagent A used in the following conversion.
\[\ce{Phenol ->[A] Benzoquinone}\]
Which of the following on oxidation yields ethyl methyl ketone?
A reaction of phenol with chloroform in presence of sodium hydroxide to form salicylaldehyde is known as ____________.
Name the catalyst used in commercial method of preparation of phenol.
Identify the compound amongst the following of which 0.1 M aqueous solution has highest boiling point.
The acid, which contains both -OH and -COOR groups is ______.
What is the action of nitrous acid on aniline?
Write the product when 1°, 2° and 3° alcohol vapours are passed over hot copper.
