Advertisements
Advertisements
प्रश्न
An organic acid X is a liquid, which often freezes during winter time in cold countries, having the molecular formula C2H4O2. On warming it with methanol in the presence of a few drops of concentrated sulphuric acid, a compound Y with a sweet smell is formed.
(a) Identify X and Y. Also write their formulae showing the functional group present in them.
(b) Write a chemical equation for the reaction involved.
उत्तर
(a) The organic acid, X, which often freezes in the winters in cold countries, is ethanoic acid. It has the molecular formula CH3COOH.
The formula with its functional group is represented as:
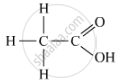
When ethanoic acid is warmed with methanol in the presence of a few drops of concentrated sulphuric acid, the compound 'Y' with sweet fragrance is formed. This compound is called methyl ethanoate, and its molecular formula is CH3COOCH3.
The formula with its functional group is represented as:
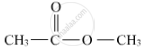
(b) The chemical equation of the above stated reaction is as follows:
`CH_3 COOH + C_2 H_5 OH` 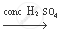 `CH_3 COOC_2 H_5 + H_2O`
`CH_3 COOC_2 H_5 + H_2O`
APPEARS IN
संबंधित प्रश्न
Complete the following chemical equations : C2H5OH`("Conc."H_2SO_4)/(443K)`>
Give the common name and IUPAC name of C2H5OH.
What happens when propanoic acid is warmed with methanol in the presence of a few drops of concentrated sulphuric acid? Write equation of the reaction involved.
What type to compound is CH3COOH?
How is acetic acid prepared from acetylene?
On adding acetic acid to sodium hydrogen carbonate in a test tube, a student observes
(A) no reaction
(B) a colourless gas with pungent smell
(C) bubbles of a colourless and odourless gas
(D) a strong smell of vinegar
The functional group present in acetic acid is ______.
Write a balanced chemical equation for the following:
A mixture of sodalime and sodium acetate is heated.
Draw the structure formula of ethyne.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
