Advertisements
Advertisements
प्रश्न
Answer the following.
Describe the structure of the Ozone. Give two uses of ozone.
उत्तर
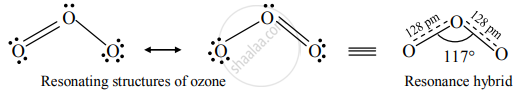
Structure of ozone:
Ozone (O3) is an angular molecule. The two O – O bond lengths in the ozone molecule are identical, 128 pm, and the O – O – O bond angle of about 117°. It is a resonance hybrid of two canonical forms.
Uses of ozone:
- Ozone is used for air purification at crowded places like cinema halls, tunnels, railways, etc.
- In sterilizing drinking water by oxidising all germs and bacteria.
- For bleaching ivory, oils, starch, wax and delicate fabrics such as silk.
- In the manufacture of synthetic camphor, potassium permanganate, etc.
APPEARS IN
संबंधित प्रश्न
Answer the following.
Name two gases which deplete ozone layer.
Answer the following.
Give one example showing reducing property of ozone.
Answer the following.
What happens when nitric oxide reacts with ozone.
Discuss the structure of sulfur dioxide.
What are interhalogen compounds?
Answer the following.
How are xenon fluorides XeF2, XeF4 and XeF6 obtained? Give suitable reactions.
Answer the following.
How are XeO3 and XeOF4 prepared?
Select the INCORRECT statement.
Which of the following molecule does not contain oxygen?
Which of the following statements is INCORRECT?
In resonance hybrid of ozone molecule, O-O bond length is ____________.
What is the colour of gaseous ozone?
Which one is amphoteric oxide?
Identify the element that forms amphoteric oxide.
What will be the formula of an oxide of iodine (atomic mass = 127) which contains 25.4 g of iodine and 8g of oxygen?
Write balanced equation of a reaction in which ozone reduces hydrogen peroxide.
Write the reaction of the following with concentrated H2SO4:
CaF2
Calculate the oxidation state of S in oleum
Mention the value of the bond angle in SO2.
Electrolytic method of preparation of dioxygen.
Industrial method of preparation of dioxygen.
