Advertisements
Advertisements
प्रश्न
Apply first law for an adiabatic process.
उत्तर
This is a process in which no heat flows into or out of the system (Q = 0). But the gas can expand by spending its internal energy or gas can be compressed through some external work. So the pressure, volume and temperature of the system may change in an adiabatic process. For an adiabatic process, the first law becomes ∆U = W.
APPEARS IN
संबंधित प्रश्न
What is a thermodynamic process?
Draw a p-V diagram showing positive work with varying pressure.
Draw a p-V diagram showing negative work with varying pressure.
Draw the PV diagram for the isobaric process.
Explain in detail an adiabatic process.
Draw the TP diagram (P-x axis, T-y axis), VT(T-x axis, V-y axis) diagram for
- Isochoric process
- Isothermal process
- Isobaric process
One mole of an ideal gas with `gamma` = 1.4 is adiabatically compressed so that its temperature rises from 27° C to 47° C. The change in the internal energy of the gas is (R = 8.3 J/mol.K) ____________.
An ideal gas A and a real gas B have their volumes increased from V to 2V under isothermal conditions. The increase in internal energy ____________.
An ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
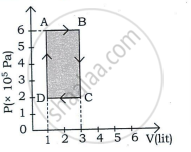
An ideal gas is taken through a cyclic process ABCDA as shown in figure. The net work done by the gas during the cycle is ______.