Advertisements
Advertisements
प्रश्न
Draw a p-V diagram showing positive work with varying pressure.
उत्तर
p-V diagram showing positive work with varying pressure:

APPEARS IN
संबंधित प्रश्न
Explain why The climate of a harbour town is more temperate than that of a town in a desert at the same latitude.
An ideal gas is taken through an isothermal process. If it does 2000 J of work on its environment, how much heat is added to it?
For work done to be reversible, the process should be ______
Draw a p-V diagram showing positive work at constant pressure.
State the assumptions made for thermodynamic processes.
Differentiate between the reversible and irreversible processes.
When a cycle tyre suddenly bursts, the air inside the tyre expands. This process is ____________.
When food is cooked in a vessel by keeping the lid closed, after some time the steam pushes the lid outward. By considering the steam as a thermodynamic system, then in the cooking process
When you exercise in the morning, by considering your body as a thermodynamic system, which of the following is true?
Apply first law for an isothermal process.
Apply first law for an adiabatic process.
Draw the PV diagram for the adiabatic process.
Draw the PV diagram for the isobaric process.
Explain in detail the isothermal process.
Explain the isobaric process and derive the work done in this process.
In a petrol engine, (internal combustion engine) air at atmospheric pressure and temperature of 20°C is compressed in the cylinder by the piston to `1/8` of its original volume. Calculate the temperature of the compressed air. (For air γ = 1.4)
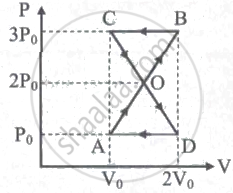
An ideal gas is taken in a cyclic process as shown in the figure. Calculate
- work done by the gas
- work done on the gas
- Net work done in the process

A thermodynamic system undergoes cyclic process ABCDA as shown in the figure. The work done by the system is ______
Two identical samples of a gas are allowed to expand (i) isothermally (ii) adiabatically. Work done is ____________.
For an isothermal expansion of a perfect gas, the value of `(Delta "P")/"P"` is equal to ____________.
Assertion: Equal volumes of monatomic and polyatomic gases are adiabatically compressed separately to equal compression ratio `("P"_2/"P"_1)`. Then monatomic gas will have greater final volume.
Reason: Among ideal gases, molecules of a monatomic gas have the smallest number of degrees of freedom.
Ideal gas for which 'ϒ' = 1.5 is suddenly compressed to `1/4`th of its initial volume. The ratio of 4 the final pressure to the initial pressure is ______.
`(ϒ = "C"_"p"/"C"_"v")`
Consider P-V diagram for an ideal gas shown in figure.
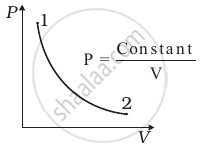
Out of the following diagrams (figure), which represents the T-P diagram?
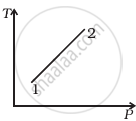 (i) |
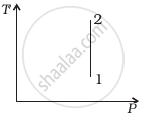 (ii) |
 (iii) |
 (iv) |
Give any two types of a thermodynamic process.
In the figure shown here, the work done in the process ACBA is ______.
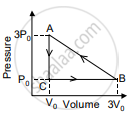
Explain the thermodynamic process.
