Advertisements
Advertisements
प्रश्न
In a petrol engine, (internal combustion engine) air at atmospheric pressure and temperature of 20°C is compressed in the cylinder by the piston to `1/8` of its original volume. Calculate the temperature of the compressed air. (For air γ = 1.4)
उत्तर
T1 = 20°C = 20 + 273 = 293 K
V1 = 1 m3
V2 = `1/8` V1m3
γ = 1.4
From equation of adiabatic process
TVγ − 1 = constant
`"T"_1"V"_1^(γ - 1) = "T"_2"V"_2^(γ - 1)`
T2 = `("V"_1/"V"_2)^(γ - 1)"T"_1`
T2 = `[1/((1/8))]^(1.4 - 1) xx 293`
= (8)0.4 × 293
= 673.1 K
= 673.1 − 273
T2 ≅ 400°C
APPEARS IN
संबंधित प्रश्न
What is a thermodynamic process?
Explain graphically (i) positive work with varying pressure, (ii) negative work with varying pressure, and (iii) positive work at constant pressure.
When food is cooked in a vessel by keeping the lid closed, after some time the steam pushes the lid outward. By considering the steam as a thermodynamic system, then in the cooking process
The V-T diagram of an ideal gas which goes through a reversible cycle A→B→C→D is shown below. (Processes D→A and B→C are adiabatic)
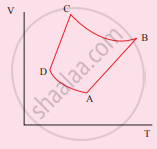
The corresponding PV diagram for the process is (all figures are schematic)
Give the equation of state for an isothermal process.
What is a cyclic process?
Explain in detail an adiabatic process.
A monoatomic gas of pressure p having volume V expands isothermally to a volume 2V and then adiabatically to a volume 16V. The final pressure of the gas is ____________.
`("ratio of specific heats" = 5/3)`
One mole of an ideal gas with `gamma` = 1.4 is adiabatically compressed so that its temperature rises from 27° C to 47° C. The change in the internal energy of the gas is (R = 8.3 J/mol.K) ____________.
Which of the following processes is reversible?
