Advertisements
Advertisements
प्रश्न
In an adiabatic expansion of the air, the volume is increased by 4%, what is the percentage change in pressure? (For air γ = 1.4)
उत्तर
From the equation for the adiabatic process,
PVγ = constant
Using differentiation, we get
Pγ Vγ − 1 dV + dP.Vγ = 0
`"dP"/"P" = -γ "dV"/"V"`
Volume 'V' is increased by 4% and γ = 1.4
`"dP"/"P" xx 100 = -γ ("dV"/"V" xx 100)`
= − 1.4 × 4
= − 5.6
Pressure is decreased by 5.6%
APPEARS IN
संबंधित प्रश्न
What is a thermodynamic process?
Write a note on free expansion.
Draw the PV diagram for the adiabatic process.
A monoatomic gas of pressure p having volume V expands isothermally to a volume 2V and then adiabatically to a volume 16V. The final pressure of the gas is ____________.
`("ratio of specific heats" = 5/3)`
An ideal gas is expanded isothermally from volume V1 to volume V2 and then compressed adiabatically to original volume V1. If the initial pressure is P1, the final pressure is P3 and net work done is W, then ____________.
One mole of an ideal gas with `gamma` = 1.4 is adiabatically compressed so that its temperature rises from 27° C to 47° C. The change in the internal energy of the gas is (R = 8.3 J/mol.K) ____________.
Two identical samples of a gas are allowed to expand (i) isothermally (ii) adiabatically. Work done is ____________.
An ideal gas A and a real gas B have their volumes increased from V to 2V under isothermal conditions. The increase in internal energy ____________.
Consider P-V diagram for an ideal gas shown in figure.
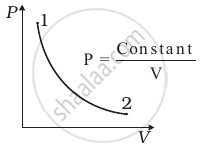
Out of the following diagrams (figure), which represents the T-P diagram?
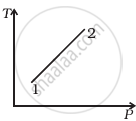 (i) |
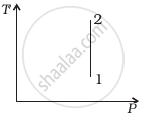 (ii) |
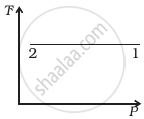 (iii) |
 (iv) |
In a certain thermodynamical process, the pressure of a gas depends on its volume as kV3. The work done when the temperature changes from 100°C to 300°C will be ______ nR, where n denotes number of moles of a gas.
