Advertisements
Advertisements
प्रश्न
Draw the PV diagram for the adiabatic process.
उत्तर

APPEARS IN
संबंधित प्रश्न
Draw a p-V diagram showing positive work at constant pressure.
Explain the cyclic process.
Apply first law for an adiabatic process.
Apply first law for an isobaric process.
What is a cyclic process?
Can the given heat energy be completely converted to work in a cyclic process? If not, when can the heat can completely converted to work?
Derive the work done in an adiabatic process.
Explain the isobaric process and derive the work done in this process.
The work done on the system in changing the state of a gas adiabatically from equilibrium state A to equilibrium state B is 22.4 J. If the gas is taken from state A to B through another process in which the net heat absorbed by the system is 15.5 cal, then the net work done by the system in the latter case is ______.
( l cal = 4.2 J)
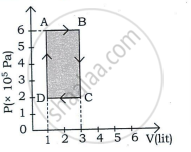
An ideal gas is taken through a cyclic process ABCDA as shown in figure. The net work done by the gas during the cycle is ______.