Advertisements
Advertisements
प्रश्न
Arrange the following compounds in increasing order of their boiling points.
(a) \[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
\backslash\phantom{.............}\\
\ce{CH - CH2Br}\\
/\phantom{.............}\\
\ce{CH3}\phantom{.................}
\end{array}\]
(b) \[\ce{CH3CH2CH2CH2Br}\]
(c) \[\begin{array}{cc}
\phantom{...}\ce{CH3}\\
\phantom{}|\\
\ce{H3C - C - CH3}\\
\phantom{}|\\
\phantom{..}\ce{Br}
\end{array}\]
पर्याय
(b) < (a) < (c)
(a) < (b) < (c)
(c) < (a) < (b)
(c) < (b) < (a)
उत्तर
(c) < (a) < (b)
Explanation:
Boiling points of isomeric haloalkanes decrease with increase in branching as with increase in branching surface area decreases which leads to decrease in intermolecular forces.
APPEARS IN
संबंधित प्रश्न
Give reasons : n-Butyl bromide has higher boiling point than t-butyl bromide.
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
Explain why alkyl halides, though polar, are immiscible with water?
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
How many structural isomers are possible for a compound with the molecular formula C3H7Cl?
Which of the following halide is 2°?
Arrange the following compounds in the increasing order of their densities.
(a)
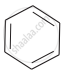
(b)
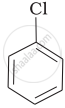
(c)
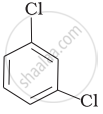
(d)
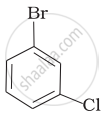
Which is the correct increasing order of boiling points of the following compounds?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Out of o-and p-dibromobenzene which one has higher melting point and why?
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
