Advertisements
Advertisements
प्रश्न
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
Why p-dichlorobenzene has higher melting point than those of ortho and meta-isomers?
p-Dichlorobenzene has higher m.p. than those of o- and m-isomers. Discuss.
उत्तर
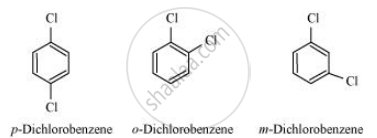
p-Dichlorobenzene is more symmetrical than o-and m-isomers. For this reason, it fits more closely than o-and m-isomers in the crystal lattice. Therefore, more energy is required to break the crystal lattice of p-dichlorobenzene. As a result, p-dichlorobenzene has a higher melting point and lower solubility than o-and m-isomers.
APPEARS IN
संबंधित प्रश्न
Give reasons : n-Butyl bromide has higher boiling point than t-butyl bromide.
Arrange the set of compounds in order of increasing boiling points.
1-Chloropropane, Isopropyl chloride, 1-Chlorobutane.
Explain why alkyl halides, though polar, are immiscible with water?
Define racemic mixture.
Why dextro and laevorotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
Which of the following compounds has the highest boiling point?
How many structural isomers are possible for a compound with the molecular formula C3H7Cl?
The decreasing order of boiling points of alkyl halides is:
Which of the following possesses the highest melting point?
Mg reacts with RBr best in ____________.
Out of o-and p-dibromobenzene which one has higher melting point and why?
Which of the following compounds will have the highest melting point and why?
| (I) |  |
|
(II) |
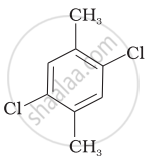 |
| (III) | 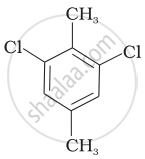 |
Which out of the following is an intensive property?
Arrange the following compounds in increasing order of their boiling points:
CH3CH2OH, CH3−CHO, CH3−COOH
Why alkyl halides though polar are immiscible with water?
Why is the boiling point of o-dichlorobenzene higher than p-dichlorobenzene, but the melting point of para-isomer is higher than ortho-isomer?
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
