Advertisements
Advertisements
प्रश्न
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
उत्तर
m-dicholrobenzene < o-dicholrobenzene < p-dicholrobenze
symmetrical structure and close packing in para isomer
ortho has a stronger dipole interaction as compared to meta
APPEARS IN
संबंधित प्रश्न
Arrange the set of compounds in order of increasing boiling points.
1-Chloropropane, Isopropyl chloride, 1-Chlorobutane.
Explain why alkyl halides, though polar, are immiscible with water?
Why dextro and laevorotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
Mg reacts with RBr best in ____________.
Arrange the following compounds in increasing order of their boiling points.
(a) \[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
\backslash\phantom{.............}\\
\ce{CH - CH2Br}\\
/\phantom{.............}\\
\ce{CH3}\phantom{.................}
\end{array}\]
(b) \[\ce{CH3CH2CH2CH2Br}\]
(c) \[\begin{array}{cc}
\phantom{...}\ce{CH3}\\
\phantom{}|\\
\ce{H3C - C - CH3}\\
\phantom{}|\\
\phantom{..}\ce{Br}
\end{array}\]
Reaction of \[\ce{C6H5CH2Br}\] with aqueous sodium hydroxide follows ______.
Which is the correct increasing order of boiling points of the following compounds?
1-Iodobutane, 1-Bromobutane, 1-Chlorobutane, Butane
Which of the following compounds will have the highest melting point and why?
| (I) |  |
|
(II) |
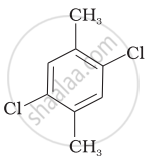 |
| (III) | 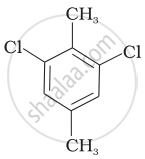 |
Why is the boiling point of o-dichlorobenzene higher than p-dichlorobenzene, but the melting point of para-isomer is higher than ortho-isomer?
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3 C(C2H5)2CH2Br}\]
