Advertisements
Advertisements
प्रश्न
Atomic number of \[\ce{Mn}\], \[\ce{Fe}\] and \[\ce{Co}\] are 25, 26 and 27 respectively. Which of the following inner orbital octahedral complex ions are diamagnetic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Mn(CN)6]^{3-}}\]
(iii) \[\ce{[Fe(CN)6]^{4-}}\]
(iv) \[\ce{[Fe(CN)6]^{3-}}\]
उत्तर
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(iii) \[\ce{[Fe(CN)6]^{4-}}\]
Explanation:
(i) Molecular orbital electronic configuration of \[\ce{Co^{3+}}\] in \[\ce{[Co(NH3)6]^{3+}}\] is
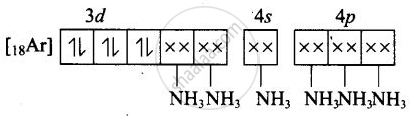
Number of unpaired electron = 0
Magnetic property = Diamagnetic
(ii) Molecular orbital electronic configuration of \[\ce{Mn^{3+}}\] in \[\ce{[Mn(CN)6]^{3-}}\]
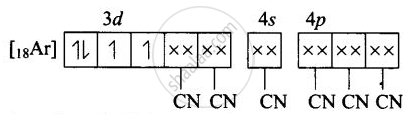
Number of unpaired electron = 2
Magnetic property = Paramagnetic
(iii) Molecular orbital electronic configuration of \[\ce{Fe^{3+}}\] in \[\ce{[Fe(CN)6]^{4-}}\] is
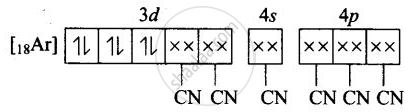
Number of unpaired electron = 0
Magnetic property = Diamagnetic
(iv) Molecular orbital electronic configuration of \[\ce{Fe^{3+}}\] in \[\ce{[Fe(CN)6]^{3-}}\]
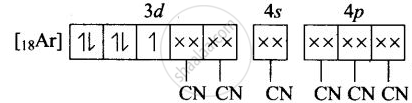
Number of unpaired electron = 1
Magnetic property = Paramagnetic
APPEARS IN
संबंधित प्रश्न
On the basis of crystal field theory, write the electronic configuration for d4 ion if ∆0 < P.
Draw figure to show the splitting of d orbitals in an octahedral crystal field.
How does the magnitude of Δ0 decide the actual configuration of d orbitals in a coordination entity?
Atomic number of \[\ce{Mn, Fe, Co}\] and Ni are 25, 26, 27 and 28 respectively. Which of the following outer orbital octahedral complexes have same number of unpaired electrons?
(i) \[\ce{[MnCl6]^{3-}}\]
(ii) \[\ce{[FeF6]^{3-}}\]
(iii) \[\ce{[CoF6]^{3-}}\]
(iv) \[\ce{[Ni(NH3)6]^{2+}}\]
Arrange following complex ions in increasing order of crystal field splitting energy (∆O):
\[\ce{[Cr(Cl)6]^{3-}, [Cr(CN)6]^{3-}, [Cr(NH3)6]^{3+}}\].
The CFSE for octahedral [CoCl6]−4 is 18,000 cm−1. What will be the CFSE for tetrahedral [CoCl3]−2?
[Ni(H2O)6]2+ (aq) is green in colour whereas [Ni(H2O)4 (en)]2+ (aq)is blue in colour, give reason in support of your answer.
Using crystal field theory, write the electronic configuration of d5 ion, if Δ0 > P.
What is crystal field splitting energy?
The complex that has highest crystal field splitting energy (Δ) is ______.
