Advertisements
Advertisements
प्रश्न
Draw figure to show the splitting of d orbitals in an octahedral crystal field.
उत्तर
Let the six ligands be symmetrically located along the Cartesian axes and the metal atom is at the origin.
On approaching the ligand, the energy of the d-orbitals increases as expected compared to the free ions. As is the case in a spherical crystal field.
The orbitals along the axes (`d_(Z^2)` and `d_(x^2 - y^2)`) repel more strongly than the dxy, dyz and dzx orbitals and have lobes directed between the axes. In the spherical crystal field, the `d_(Z^2)` and `d_(x^2 - y^2)` orbitals increase in energy and the dxy, dyz, dzx orbitals decrease in energy compared to the average energy.
Therefore, the degenerate set of d-orbitals splits into two groups – the low energy orbital group t2g and the high energy orbital group eg separated by energy Δ0.
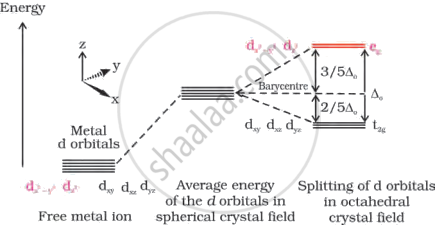
d orbital splitting in an octahedral crystal field
APPEARS IN
संबंधित प्रश्न
On the basis of crystal field theory, write the electronic configuration for d4 ion if ∆0 < P.
The hexaquo manganese (II) ion contains five unpaired electrons, while the hexacyanoion contains only one unpaired electron. Explain using Crystal Field Theory.
How does the magnitude of Δ0 decide the actual configuration of d orbitals in a coordination entity?
State the superiority of crystal field theory over valence bond theory.
Why are low spin tetrahedral complexes rarely observed?
Write the electronic configuration of Fe(III) on the basis of crystal field theory when it forms an octahedral complex in the presence of (i) strong field, and (ii) weak field ligand. (Atomic no.of Fe=26)
Draw the structures of the following :
(1) XeF6
(2) IF7
Complete and balance the following reactions:
(1) P4 + H2SO4 → ____ + _____ + _____ + _____
(2) Ag + HNO3(dilute) → _____ + ______ + _____ + _____
The CFSE for octahedral \[\ce{[CoCl6]^{4-}}\] is 18,000 cm–1. The CFSE for tetrahedral \[\ce{[CoCl4]^{2-}}\] will be ______.
On the basis of crystal field theory explain why Co(III) forms paramagnetic octahedral complex with weak field ligands whereas it forms diamagnetic octahedral complex with strong field ligands.
Using crystal field theory, draw energy level diagram, write electronic configuration of the central metal atom/ion and determine the magnetic moment value in the following:
\[\ce{[FeF6]^{3-}, [Fe(H2O)6]^{2+}, [Fe(CN)6]^{4-}}\]
The CFSE for octahedral [CoCl6]−4 is 18,000 cm−1. What will be the CFSE for tetrahedral [CoCl3]−2?
[Ni(H2O)6]2+ (aq) is green in colour whereas [Ni(H2O)4 (en)]2+ (aq)is blue in colour, give reason in support of your answer.
In a coordination entity, the electronic configuration of the central metal ion is t2g3 eg1
Draw the crystal field splitting diagram for the above complex.
The correct order of increasing crystal field strength in following series:
The CFSE of [CoCl6]3– is 18000 cm–1 the CFSE for [CoCl4]– will be ______.
What is the difference between a weak field ligand and a strong field ligand?
On the basis of crystal field theory, write the electronic configuration for the d5 ion with a weak ligand for which Δ0 < P.
Explain the difference between a weak field ligand and a strong field ligand.
