Advertisements
Advertisements
प्रश्न
Attempt the following:
Write preparation, properties and uses of Teflon.
उत्तर
Preparation of teflon:
- The monomer used in preparation of teflon is tetrafluoroethylene, (CF2 = CF2), which is a gas at room temperature.
- Tetrafluoroethylene is polymerized by using free-radical initiators such as hydrogen peroxide or ammonium persulphate at high pressure to produce polytetrafluoroethylene (teflon).
\[\ce{\underset{Tetrafluoroethylene}{{n} CF2 = CF2}->[Polymerization][Peroxide]\underset{Teflon}{[-CF2 - CF2 -]}_{n}}\]
Properties of teflon:
- Teflon is tough, chemically inert and resistant to heat and attack by corrosive reagents.
- C – F bond is very difficult to break and remains unaffected by corrosive alkali, organic solvents.
Uses: Teflon is used in making non-stick cookware, oil seals, gaskets, etc.
संबंधित प्रश्न
Write any ‘two' uses of terylene.
Write the chemical reaction to prepare novolac polymer.
The Zieglar-Natta catalyst is used in the preparation of _______.
(A) LDPE
(B) PHBV
(C) PAN
(D) HDPE
Choose the correct option from the given alternatives.
Which of the following is made up of polyamides?
Answer the following in one sentence.
Define the term: Vulcanization
Answer the following.
Match the following pairs:
| Name of polymer | Monomer |
| 1. Teflon | a. CH2 = CH2 |
| 2. PVC | b. CF2 = CF2 |
| 3. Polyester | c. CH2 = CHCl |
| 4. Polythene | d. C6H5OH and HCHO |
| 5. Bakelite | e. Dicarboxylic acid and polyhydoxyglycol |
Answer the following.
Write name and formula of raw material from which bakelite is made.
Identify condensation polymers and addition polymers from the following.
-(CO(CH2)4 - CONH(CH2)6NH-)n
Identify condensation polymers and addition polymers from the following.
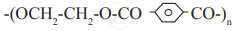
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Write the reaction involved in the formation of:
Bakelite
Nylon 6, 6 is a condensation polymer of hexamethylenediamine and _____________
Write the name of the catalyst used for preparation of high density polythene polymer.
Monomer used for preparation of polyacrylonitrile is _____________
Write a chemical reaction for the preparation of the following polymer.
polyacrylonitrile
Write preparation of low density polythene.
Mention two uses of LDP.
Write chemical reaction for preparation of the following.
Buna-S
Write chemical reaction for preparation of the following.
Neoprene
Which among the following polymers is obtained from styrene and 1-3-butadiene?
Which among the following polymers is used for making handles of cooker?
How many isoprene units are present in abscisic acid?
Identify the CORRECT statement regarding the following polymer.
\[\begin{array}{cc}
\phantom{....}\ce{O}\phantom{............}\ce{O}\phantom{...................}\ce{H}\phantom{.....}\\
\phantom{....}||\phantom{.............}||\phantom{...................}|\phantom{......}\\
\ce{-[C - (CH2)4 - C - NH - (CH2)6 - N -]_{{n}}}
\end{array}\]
The INCORRECT match for the polymer with its application is:
Identify the INCORRECT match.
\[\ce{{n} CH2 = CH2 ->[333 K - 343 K][6 - 7 atm, catalyst] X}\]
Which of the following is CORRECT about polymer 'X'?
Which of the following polymers is obtained from chloroprene?
Identify the polymer obtained by polymerization of n moles of acrylonitrile.
Which of the following compounds is used to prepare orlon?
Which among the following polymers is obtained from CH2 = CH – CN by polymerisation?
Which of the following monomers is used in the manufacture of Neoprene rubber?
Which among the following polymers is used to manufacture chemical containers?
The commercial name of polyacrylonitrile is ______.
The monomer of Teflon is ______.
F2C = CF2 is monomer of the polymer -
Which of the following polymer has ester linkage?
Trans - form of poly isoprene is:-
Which of the following is an example of polyester?
Which of the following polymers is synthesized using a free radical polymerisation technique?
Nylon threads are made of ______.
The catalyst used for the polymerisation of olefins is ______.
Which among the following polymers has high tensile strength and is used to obtain tyre cords?
Identify the monomer used to prepare neoprene.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the name of the monomer of natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
