Advertisements
Advertisements
प्रश्न
Can a system be heated and its temperature remains constant?
उत्तर
If the system utilises the entire heat energy supplied in doing the work against the surroundings, then there will be no change in the internal energy of the system and the temperature will remain constant.
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Which of the following is an example of the first law of thermodynamics?
For an Isochoric process
Define an isolated system.
Calculate the amount of work done during isothermal expansion of a gas from a volume of 4 dm3 to 6 dm3 against a constant external pressure of 3 atmosphere?
Is it possible to increase the temperature of a gas without adding heat to it? Explain.
Air pressure in a car tyre increases during driving. Explain.
A system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is ______.
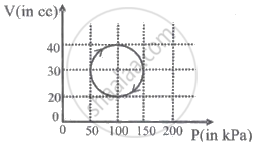
ΔU = 0 is true for ______.
The amount of work done in increasing the voltage across the plates of capacitor from 5 V to 10 V is W. The work done in increasing it from 10 V to 15 V will be ______.
Define the isothermal process.
