Advertisements
Advertisements
प्रश्न
Choose the correct option.
Which of the following is an example of the first law of thermodynamics?
पर्याय
The specific heat of an object explains how easily it changes temperatures.
While melting, an ice cube remains at the same temperature.
When a refrigerator is unplugged, everything inside of it returns to room temperature after some time.
After falling down the hill, a ball's kinetic energy plus heat energy equals the initial potential energy.
उत्तर
After falling down the hill, a ball's kinetic energy plus heat energy equals the initial potential energy.
Explanation:
There are three laws of thermodynamics. These laws explain the relationship between quantities such as heat, temperature, entropy, and work. The first law of thermodynamics defines that for the change in the internal energy, which becomes equal to the heat that adds to the system minus the work which is done by the system.
The expression obtained is,
ΔU = Q – W
Here, ΔU is the change in internal energy.
Q is the heat added to the system.
W is the work done by the system.
The second law of thermodynamics defines that the total entropy in an isolated system will never decrease, and this is a constant only if all the process is reversible.
The third law of thermodynamics defines that the entropy in a system at the absolute zero temperature is a constant. Because the system here at zero temperature gets exists in the ground state.
APPEARS IN
संबंधित प्रश्न
A system can be taken from the initial state p1, V1 to the final state p2, V2 by two different methods. Let ∆Q and ∆W represent the heat given to the system and the work done by the system. Which of the following must be the same in both the methods?
The internal energy of an ideal gas decreases by the same amount as the work done by the system.
(a) The process must be adiabatic.
b) The process must be isothermal.
(c) The process must be isobaric.
(d) The temperature must decrease.
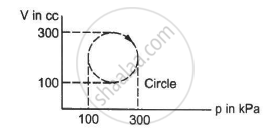
Calculate the heat absorbed by a system in going through the cyclic process shown in figure.
The internal energy of a gas is given by U = 1.5 pV. It expands from 100 cm3 to 200 cm3against a constant pressure of 1.0 × 105 Pa. Calculate the heat absorbed by the gas in the process.
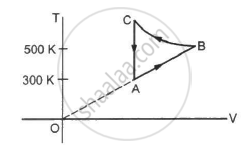
Consider the cyclic process ABCA, shown in figure, performed on a sample of 2.0 mol of an ideal gas. A total of 1200 J of heat is withdrawn from the sample in the process. Find the work done by the gas during the part BC.
For an Isothermal process
For an Isochoric process
When heat energy of 2000 joules is supplied to a gas at constant pressure 2.1 x 105 N/m2, there is an increase in its volume equal to 2.5 x 10-3 m3. The increase in internal energy of the gas in joules is ____________.
The process, in which no heat enters or leaves the system, is termed as ____________.
Three copper blocks of masses M1, M2 and M3 kg respectively are brought into thermal contact till they reach equilibrium. Before contact, they were at T1, T2, T3 (T1 > T2 > T3). Assuming there is no heat loss to the surroundings, the equilibrium temprature T is (s is specific heat of copper)
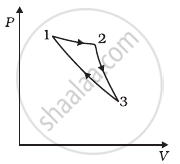
Consider a cycle followed by an engine (Figure)
1 to 2 is isothermal
2 to 3 is adiabatic
3 to 1 is adiabatic
Such a process does not exist because ______.
- heat is completely converted to mechanical energy in such a process, which is not possible.
- mechanical energy is completely converted to heat in this process, which is not possible.
- curves representing two adiabatic processes don’t intersect.
- curves representing an adiabatic process and an isothermal process don’t intersect.
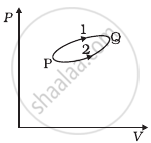
A system goes from P to Q by two different paths in the P-V diagram as shown in figure. Heat given to the system in path 1 is 1000 J. The work done by the system along path 1 is more than path 2 by 100 J. What is the heat exchanged by the system in path 2?
A cycle followed by an engine (made of one mole of perfect gas in a cylinder with a piston) is shown in figure.
A to B : volume constant
B to C : adiabatic
C to D : volume constant
D to A : adiabatic
VC = VD = 2VA = 2VB

- In which part of the cycle heat is supplied to the engine from outside?
- In which part of the cycle heat is being given to the surrounding by the engine?
- What is the work done by the engine in one cycle? Write your answer in term of PA, PB, VA.
- What is the efficiency of the engine?
(γ = `5/3` for the gas), (Cv = `3/2` R for one mole)
A cycle followed by an engine (made of one mole of an ideal gas in a cylinder with a piston) is shown in figure. Find heat exchanged by the engine, with the surroundings for each section of the cycle. (Cv = (3/2)R)
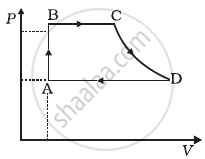
- AB : constant volume
- BC : constant pressure
- CD : adiabatic
- DA : constant pressure
Consider that an ideal gas (n moles) is expanding in a process given by P = f(V), which passes through a point (V0, P0). Show that the gas is absorbing heat at (P0, V0) if the slope of the curve P = f(V) is larger than the slope of the adiabat passing through (P0, V0).
Write the mathematical equation for the first law of thermodynamics for:
Isothermal process
Write the mathematical equation for the first law of thermodynamics for:
Adiabatic process
An electric appliance supplies 6000 J/min heat to the system. If the system delivers a power of 90 W. How long it would take to increase the internal energy by 2.5 × 103 J?
An ideal gas is taken through series of changes ABCA. The amount of work involved in the cycle is ______.
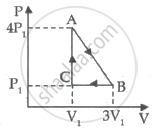
Mathematical equation of first law of thermodynamics for isochoric process is ______.
ΔU = 0 is true for ______.
One mole of an ideal gas is initially kept in a cylinder with a movable frictionless and massless piston at pressure of 1.01MPa, and temperature 27°C. It is then expanded till its volume is doubled. How much work is done if the expansion is isobaric?
If the adiabatic ratio for a gas is 5/3, find the molar specific heat capacity of the gas at (i) constant volume (ii) constant pressure.
What is true for an adiabatic process?
For an isothermal and reversible expansion of 0.5 mol of an ideal gas Wmax is - 3.918 kJ. The value of ΔU is ______.
Using the first law of thermodynamics, show that for an ideal gas, the difference between the molar specific heat capacities at constant pressure and at constant volume is equal to the molar gas constant R.
Consider the cyclic process ABCA on a sample of 2.0 mol of an ideal gas as shown in following figure. The temperature of the gas at A and B are 300 K and 500 K respectively. A total of 1200 J heat is withdrawn from the sample in this process. Find the work done by the gas in part BC. (R = 8.3 J/mol K)
Obtain an expression for the workdone by a gas in an isothermal process.
