Advertisements
Advertisements
प्रश्न
A cycle followed by an engine (made of one mole of an ideal gas in a cylinder with a piston) is shown in figure. Find heat exchanged by the engine, with the surroundings for each section of the cycle. (Cv = (3/2)R)
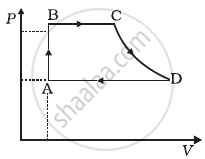
- AB : constant volume
- BC : constant pressure
- CD : adiabatic
- DA : constant pressure
उत्तर
a. By using the first law of thermodynamics, we can find the amount of heat associated with each process
For process AB
Volume is constant, hence work done dW = 0
According to first law of thermodynamics,
`ΔQ = ΔU + ΔW = ΔU + 0 = ΔU`
= `nCvΔT = nCv(T_B - T_A)`
= `3/2 R(T_B - T_A)` .....(∵ n = 1)
= `3/2 (RT_B - RT_A)`
= `3/2 (P_AV_B - P-AV_A)`
= `3/2 (P_B - P_A)V_A` .....[∵ VB = VA]
b. For process BC, P = constant
`ΔQ = ΔU + ΔW`
= `3/2 R(T_C - T_B) + P_B(V_C - V_B)`
= `3/2 (P_CV_C - P+BV_B) + P_B (V_C - V_B)`
= `5/2 P_B (V_C - V_B)`
Heat exchanged = `5/2 P_B (V_C - V_A)` .....(∵ PB = PC and PB = VA)
c. For process CD, QCD = 0 .....(As the change is adiabatic.)
d. In process DA involves compression of gas from VD to VA at constant pressure PA.
∴ Heat exchanged can be calculated in a similar way as process BC.
Hence, `ΔQ = 5/2 P_A(V_A - V_D)`.
APPEARS IN
संबंधित प्रश्न
Write the mathematical expression of the First Law of Thermodynamics for Isothermal Process
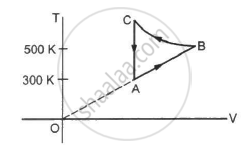
Refer to figure. Let ∆U1 and ∆U2 be the change in internal energy in processes A and B respectively, ∆Q be the net heat given to the system in process A + B and ∆W be the net work done by the system in the process A + B.

(a) ∆U1 + ∆U2 = 0
(b) ∆U1 − ∆U2 = 0
(c) ∆Q − ∆W = 0
(d) ∆Q + ∆W = 0
A thermally insulated, closed copper vessel contains water at 15°C. When the vessel is shaken vigorously for 15 minutes, the temperature rises to 17°C. The mass of the vessel is 100 g and that of the water is 200 g. The specific heat capacities of copper and water are 420 J kg−1 K−1 and 4200 J kg−1 K−1 respectively. Neglect any thermal expansion. (a) How much heat is transferred to the liquid-vessel system? (b) How much work has been done on this system? (c) How much is the increase in internal energy of the system?
Consider the cyclic process ABCA, shown in figure, performed on a sample of 2.0 mol of an ideal gas. A total of 1200 J of heat is withdrawn from the sample in the process. Find the work done by the gas during the part BC.
Define an isolated system.
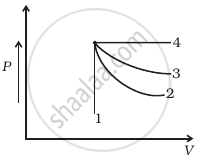
An ideal gas undergoes four different processes from the same initial state (figure). Four processes are adiabatic, isothermal, isobaric and isochoric. Out of 1, 2, 3 and 4 which one is adiabatic.
Consider one mole of perfect gas in a cylinder of unit cross section with a piston attached (figure). A spring (spring constant k) is attached (unstretched length L) to the piston and to the bottom of the cylinder. Initially the spring is unstretched and the gas is in equilibrium. A certain amount of heat Q is supplied to the gas causing an increase of volume from V0 to V1.

- What is the initial pressure of the system?
- What is the final pressure of the system?
- Using the first law of thermodynamics, write down a relation between Q, Pa, V, Vo and k.
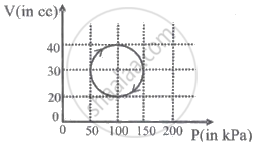
A system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is ______.
The first law of thermodynamics for isothermal process is ______.
A given system undergoes a change in which the work done by the system equals the decrease in its internal energy. The system must have undergone an ______.
