Advertisements
Advertisements
प्रश्न
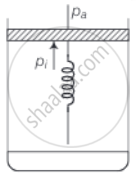
Consider one mole of perfect gas in a cylinder of unit cross section with a piston attached (figure). A spring (spring constant k) is attached (unstretched length L) to the piston and to the bottom of the cylinder. Initially the spring is unstretched and the gas is in equilibrium. A certain amount of heat Q is supplied to the gas causing an increase of volume from V0 to V1.

- What is the initial pressure of the system?
- What is the final pressure of the system?
- Using the first law of thermodynamics, write down a relation between Q, Pa, V, Vo and k.
उत्तर
a. Initially the piston is in equilibrium hence, Pf = Pa
b. On supplying heat, the gas expands from V0 to V1
∴ Increase in volume of the gas = V1 – V0
As the piston is of the unit cross-sectional area hence, extension in the spring
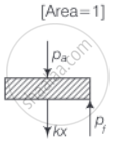
`x = (V_1 - V_0)/"Area" = V_1 - V_0`
∴ Force exerted by the spring on the piston
= `F = kx = k(V_1 - V_0)`
Hence, Final pressure = `P_f = P_a + kx`
= `P_a + k xx (V_1 - V_0)`
c. From the first law of thermodynamics `dQ = du + dW`
If T is the final temperature of the gas. then increases in internal energy
`dU = C_v (T - T_0) = C_v (T - T_0)`
We can write, `T = (P_f V_1)/R - [(P_a + k(V_1 - V_0))/R] V_1/R`
Work done by the gas = PdV + increase in PE of the spring
= `P_a (V_1 - V_0) + 1/2 kx^2`
Now, we can write `dQ = dU + dW`
= `C_V (T - T_0) + P_a (V - V_0) + 1/2 kx^2`
= `C_V (T - T_0) + P_a (V_ V_0) + 1/2 (V_1 - V_0)^2`
This is the required relation.
APPEARS IN
संबंधित प्रश्न
The pressure of a gas changes linearly with volume from 10 kPa, 200 cc to 50 kPa, 50 cc. (a) Calculate the work done by the gas. (b) If no heat is supplied or extracted from the gas, what is the change in the internal energy of the gas?
For an Isochoric process
"The mass and energy both are conserved in an isolated system", is the statement of ______.
Three copper blocks of masses M1, M2 and M3 kg respectively are brought into thermal contact till they reach equilibrium. Before contact, they were at T1, T2, T3 (T1 > T2 > T3). Assuming there is no heat loss to the surroundings, the equilibrium temprature T is (s is specific heat of copper)
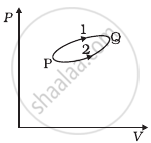
A system goes from P to Q by two different paths in the P-V diagram as shown in figure. Heat given to the system in path 1 is 1000 J. The work done by the system along path 1 is more than path 2 by 100 J. What is the heat exchanged by the system in path 2?
Write the mathematical equation for the first law of thermodynamics for:
Isothermal process
Write the mathematical equation for the first law of thermodynamics for:
Adiabatic process
An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume V1 and contains ideal gas at pressure P1 and temperature T1. The other chamber has volume V2 and contains ideal gas at pressure P2 and temperature T2. If the partition is removed without doing any work on the gas, the final equilibrium temperature of the gas in the container will be ______.
The V cc volume of gas having `γ = 5/2` is suddenly compressed to `(V/4)` cc. The initial pressure of the gas is p. The final pressure of the gas will be ______.
A monoatomic gas at 27°C is adiabatically compressed to 80% of its initial volume. Find the final temperature of the gas.
