Advertisements
Advertisements
प्रश्न
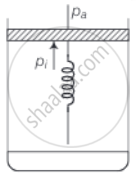
Consider one mole of perfect gas in a cylinder of unit cross section with a piston attached (figure). A spring (spring constant k) is attached (unstretched length L) to the piston and to the bottom of the cylinder. Initially the spring is unstretched and the gas is in equilibrium. A certain amount of heat Q is supplied to the gas causing an increase of volume from V0 to V1.

- What is the initial pressure of the system?
- What is the final pressure of the system?
- Using the first law of thermodynamics, write down a relation between Q, Pa, V, Vo and k.
उत्तर
a. Initially the piston is in equilibrium hence, Pf = Pa
b. On supplying heat, the gas expands from V0 to V1
∴ Increase in volume of the gas = V1 – V0
As the piston is of the unit cross-sectional area hence, extension in the spring
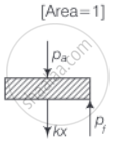
`x = (V_1 - V_0)/"Area" = V_1 - V_0`
∴ Force exerted by the spring on the piston
= `F = kx = k(V_1 - V_0)`
Hence, Final pressure = `P_f = P_a + kx`
= `P_a + k xx (V_1 - V_0)`
c. From the first law of thermodynamics `dQ = du + dW`
If T is the final temperature of the gas. then increases in internal energy
`dU = C_v (T - T_0) = C_v (T - T_0)`
We can write, `T = (P_f V_1)/R - [(P_a + k(V_1 - V_0))/R] V_1/R`
Work done by the gas = PdV + increase in PE of the spring
= `P_a (V_1 - V_0) + 1/2 kx^2`
Now, we can write `dQ = dU + dW`
= `C_V (T - T_0) + P_a (V - V_0) + 1/2 kx^2`
= `C_V (T - T_0) + P_a (V_ V_0) + 1/2 (V_1 - V_0)^2`
This is the required relation.
APPEARS IN
संबंधित प्रश्न
Answer the following in one or two sentences.
State the first law of thermodynamics.
"The mass and energy both are conserved in an isolated system", is the statement of ______.
A gas performs 0.320 kJ work on surrounding and absorbs 120 J of heat from the surrounding. Hence, change in internal energy is ______.
Consider two containers A and B containing identical gases at the same pressure, volume and temperature. The gas in container A is compressed to half of its original volume isothermally while the gas in container B is compressed to half of its original value adiabatically. The ratio of final pressure of gas in B to that of gas in A is ______.
Consider a cycle tyre being filled with air by a pump. Let V be the volume of the tyre (fixed) and at each stroke of the pump ∆V(V) of air is transferred to the tube adiabatically. What is the work done when the pressure in the tube is increased from P1 to P2?
Mathematical equation of first law of thermodynamics for isochoric process is ______.
What work will be done, when 3 moles of an ideal gas are compressed to half the initial volume at a constant temperature of 300 K?
An ideal gas having pressure p, volume V and temperature T undergoes a thermodynamic process in which dW = 0 and dQ < 0. Then, for the gas ______.
What is Isobaric process?
Define isochoric process
