Advertisements
Advertisements
प्रश्न
Consider a cycle tyre being filled with air by a pump. Let V be the volume of the tyre (fixed) and at each stroke of the pump ∆V(V) of air is transferred to the tube adiabatically. What is the work done when the pressure in the tube is increased from P1 to P2?
उत्तर
Let, volume is increased by ∆V and pressure is increased by ∆P by a stroke.
Just before and after a stroke, we can write
`P_1V_1^γ = P_2V_2^γ`
⇒ `P(V + ∆V)^γ = (P + ∆P)V^γ` .....(∵ Volume is fixed)
⇒ `PV^γ (1 + (∆V)/V)^γ = P(1 + (∆P)/P)V^γ`
⇒ `PV^γ (1 + γ (∆V)/V)^γ = PV^γ (1 + (∆P)/P)` .....(∵ ∆V << V)
⇒ `γ (∆V)/V = (∆P)/P`
⇒ `∆V = 1/γ V/P ∆P`
⇒ `dV = 1/γ V/P dP`
Hence, work done is increasing the pressure from P1 to P2
`W = int_(P_1)^(P_2) PdV`
= `int_(P_1)^(P_2) P xx 1/γ V/P dP`
= `V/γ int_(P_1)^(P_2)`
= `V/γ (P_2 - P_1)`
⇒ `W = ((P_2 - P_1))/γ V`
APPEARS IN
संबंधित प्रश्न
10 kg of four different gases (Cl2, CH4, O2, N2) expand isothermally and reversibly from 20 atm to 10 atm. The order of amount of work will be ____________.
A sample of gas absorbs 4000 kJ of heat and surrounding does 2000 J of work on sample. What is the value of ∆U?
Calculate the amount of work done during isothermal expansion of a gas from a volume of 4 dm3 to 6 dm3 against a constant external pressure of 3 atmosphere?
Change in internal energy, when 4 KJ of work is done on the system and 1 KJ heat is given out by the system, is:
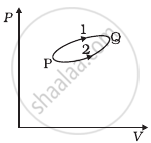
A system goes from P to Q by two different paths in the P-V diagram as shown in figure. Heat given to the system in path 1 is 1000 J. The work done by the system along path 1 is more than path 2 by 100 J. What is the heat exchanged by the system in path 2?
A cycle followed by an engine (made of one mole of an ideal gas in a cylinder with a piston) is shown in figure. Find heat exchanged by the engine, with the surroundings for each section of the cycle. (Cv = (3/2)R)
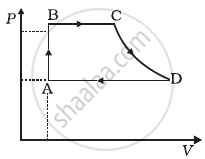
- AB : constant volume
- BC : constant pressure
- CD : adiabatic
- DA : constant pressure
ΔU = 0 is true for ______.
What is true for an adiabatic process?
Explain the formulation of first law of thrmodynamics.
Choose the correct relation with reason.
