Advertisements
Advertisements
प्रश्न
Consider a cycle tyre being filled with air by a pump. Let V be the volume of the tyre (fixed) and at each stroke of the pump ∆V(V) of air is transferred to the tube adiabatically. What is the work done when the pressure in the tube is increased from P1 to P2?
उत्तर
Let, volume is increased by ∆V and pressure is increased by ∆P by a stroke.
Just before and after a stroke, we can write
`P_1V_1^γ = P_2V_2^γ`
⇒ `P(V + ∆V)^γ = (P + ∆P)V^γ` .....(∵ Volume is fixed)
⇒ `PV^γ (1 + (∆V)/V)^γ = P(1 + (∆P)/P)V^γ`
⇒ `PV^γ (1 + γ (∆V)/V)^γ = PV^γ (1 + (∆P)/P)` .....(∵ ∆V << V)
⇒ `γ (∆V)/V = (∆P)/P`
⇒ `∆V = 1/γ V/P ∆P`
⇒ `dV = 1/γ V/P dP`
Hence, work done is increasing the pressure from P1 to P2
`W = int_(P_1)^(P_2) PdV`
= `int_(P_1)^(P_2) P xx 1/γ V/P dP`
= `V/γ int_(P_1)^(P_2)`
= `V/γ (P_2 - P_1)`
⇒ `W = ((P_2 - P_1))/γ V`
APPEARS IN
संबंधित प्रश्न
Write the mathematical expression of the First Law of Thermodynamics for Isothermal Process
The internal energy of an ideal gas decreases by the same amount as the work done by the system.
(a) The process must be adiabatic.
b) The process must be isothermal.
(c) The process must be isobaric.
(d) The temperature must decrease.
Figure shows the variation in the internal energy U with the volume V of 2.0 mol of an ideal gas in a cyclic process abcda. The temperatures of the gas at b and c are 500 K and 300 K respectively. Calculate the heat absorbed by the gas during the process.

10 kg of four different gases (Cl2, CH4, O2, N2) expand isothermally and reversibly from 20 atm to 10 atm. The order of amount of work will be ____________.
The compressibility of water is 5 × 10-10 m2/N. Pressure of 15 × 106 Pa is applied on 100 ml volume of water. The change in the volume of water is ______.
If an average person jogs, hse produces 14.5 × 103 cal/min. This is removed by the evaporation of sweat. The amount of sweat evaporated per minute (assuming 1 kg requires 580 × 103 cal for evaparation) is ______.
Consider a P-V diagram in which the path followed by one mole of perfect gas in a cylindrical container is shown in figure.
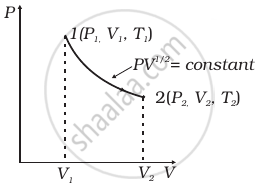
- Find the work done when the gas is taken from state 1 to state 2.
- What is the ratio of temperature T1/T2, if V2 = 2V1?
- Given the internal energy for one mole of gas at temperature T is (3/2) RT, find the heat supplied to the gas when it is taken from state 1 to 2, with V2 = 2V1.
In an adiabatic process, ______.
If the adiabatic ratio for a gas is 5/3, find the molar specific heat capacity of the gas at (i) constant volume (ii) constant pressure.
In an adiabatic process, W = ______.
