Advertisements
Advertisements
Question
Consider a cycle tyre being filled with air by a pump. Let V be the volume of the tyre (fixed) and at each stroke of the pump ∆V(V) of air is transferred to the tube adiabatically. What is the work done when the pressure in the tube is increased from P1 to P2?
Solution
Let, volume is increased by ∆V and pressure is increased by ∆P by a stroke.
Just before and after a stroke, we can write
`P_1V_1^γ = P_2V_2^γ`
⇒ `P(V + ∆V)^γ = (P + ∆P)V^γ` .....(∵ Volume is fixed)
⇒ `PV^γ (1 + (∆V)/V)^γ = P(1 + (∆P)/P)V^γ`
⇒ `PV^γ (1 + γ (∆V)/V)^γ = PV^γ (1 + (∆P)/P)` .....(∵ ∆V << V)
⇒ `γ (∆V)/V = (∆P)/P`
⇒ `∆V = 1/γ V/P ∆P`
⇒ `dV = 1/γ V/P dP`
Hence, work done is increasing the pressure from P1 to P2
`W = int_(P_1)^(P_2) PdV`
= `int_(P_1)^(P_2) P xx 1/γ V/P dP`
= `V/γ int_(P_1)^(P_2)`
= `V/γ (P_2 - P_1)`
⇒ `W = ((P_2 - P_1))/γ V`
APPEARS IN
RELATED QUESTIONS
Write the mathematical expression of the First Law of Thermodynamics for Isothermal Process
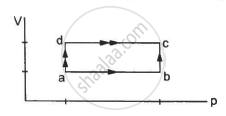
When a system is taken through the process abc shown in figure, 80 J of heat is absorbed by the system and 30 J of work is done by it. If the system does 10 J of work during the process adc, how much heat flows into it during the process?
Calculate the increase in the internal energy of 10 g of water when it is heated from 0°C to 100°C and converted into steam at 100 kPa. The density of steam = 0.6 kg m−3. Specific heat capacity of water = 4200 J kg−1 °C−1 and the latent heat of vaporization of water = 2.25 × 10 6J kg−1.
Answer the following in one or two sentences.
State the first law of thermodynamics.
A sample of gas absorbs 4000 kJ of heat and surrounding does 2000 J of work on sample. What is the value of ∆U?
Which of the following are TRUE for a reversible isothermal process?
(i) ∆U = 0
(ii) ∆H = 0
(iii) Q = W
(iv) ∆T = 0
The initial state of a certain gas is (Pi, Vi, Ti). It undergoes expansion till its volume becomes Vf. Consider the following two cases:
- the expansion takes place at constant temperature.
- the expansion takes place at constant pressure.
Plot the P-V diagram for each case. In which of the two cases, is the work done by the gas more?
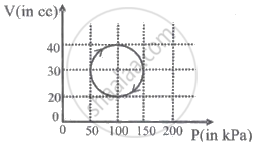
A system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is ______.
Derive an expression for the work done during an isothermal process.
What is true for an adiabatic process?
