Advertisements
Advertisements
प्रश्न
Carbon monoxide reacts with hydrogen under certain conditions to form methanol (CH3OH). Write a balanced chemical equation for this reaction indicating the physical states of reactants and product as well as the conditions under which this reaction takes place.
उत्तर
Carbon monoxide reacts with hydrogen under the following conditions:
(i) 300 atm. pressure
(ii) 300oC temperature
(iii) in the presence of ZnO and CrO3 as catalysts
The balanced chemical equation for the given reaction is as follows:
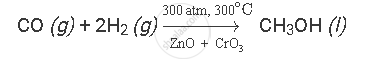
APPEARS IN
संबंधित प्रश्न
Write balanced chemical equation from the following information:
An aqueous calcium hydroxide solution (lime water) reacts with carbon dioxide gas to produce a solid calcium carbonate precipitate and water.
State the various characteristics of chemical reactions.
Balance the following equation. Also name the product formed.
FeCI2 + CI2 → FeCI3
Balance the equation stepwise.
Ag(s) + HCl(aq) → AgCl ↓+ H2 ↑
Write the chemical equation for the following word equation and balance them.
Nitrogen + Oxygen → Nitrogen monoxide
What is the valency of :
carbon in CH4
Write word equation for the following molecular equation:
Na2SO3 + 2HCl [dil.] → 2NaCl + H2O + SO2 [g]
Name the following:
Two non-metal which react explosively when brought in close contact.
Balance the following simple equation:
Zn + HCl → ZnCl2 + H2
Balance the following simple equation:
NaOH + Cl2 → NaCl + NaClO + H2O
