Advertisements
Advertisements
प्रश्न
Carbon monoxide reacts with hydrogen under certain conditions to form methanol (CH3OH). Write a balanced chemical equation for this reaction indicating the physical states of reactants and product as well as the conditions under which this reaction takes place.
उत्तर
Carbon monoxide reacts with hydrogen under the following conditions:
(i) 300 atm. pressure
(ii) 300oC temperature
(iii) in the presence of ZnO and CrO3 as catalysts
The balanced chemical equation for the given reaction is as follows:
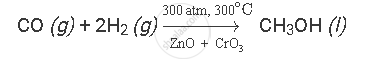
APPEARS IN
संबंधित प्रश्न
Why should a magnesium ribbon be cleaned before it is burnt in air?
Balance the given equation:
NH3 + CuO  Cu +N2 +H2O
Cu +N2 +H2O
State one characteristic of the chemical reaction which takes place when lemon juice is added gradually to potassium permanganate solution.
Show the formation of Na2O by the transfer of electrons.
Write the balanced chemical equations of the following reactions.
chlorine + sulphur dioxide + water → sulphuric acid + hydrogen chloride
Write the balanced chemical equation of the following reaction.
barium chloride + sulphuric acid → barium sulphate + hydrochloric acid
Write the balanced chemical equation of the following reaction.
aluminium carbide + water → aluminium hydroxide + methane
Write the name and molecular formula of a carbon compound having its name suffixed with "-ol" and having two carbon atoms in its molecule. With the help of a chemical equation indicate what happens when this compound is heated with excess conc. H2SO4.
Balance the following simple equation:
C + O2 → CO
Balance the following simple equation:
Fe + H2O ⇌ Fe3O4 + H2
