Advertisements
Advertisements
प्रश्न
Choose the most correct option.
Which of the following complexes exist as cis and trans isomers?
1. [Cr(NH3)2Cl4]-
2. [Co(NH3)5Br]2⊕
3. [PtCl2Br2]2- (square planar)
4. [FeCl2(NCS)2]2- (tetrahedral)
पर्याय
1 and 3
2 and 3
1 and 3
4 only
उत्तर
1 and 3
APPEARS IN
संबंधित प्रश्न
Out of 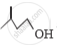 and
and 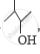 , which one is optically active and why ?
, which one is optically active and why ?
Answer the following question.
Draw isomers of the following
[Cr(en2)Br2]⊕
Draw the geometrical isomers of the following complexes [Pt(NH3)(H2O)Cl2].
Which one of the following will give a pair of enantiomorphs?
Which type of isomerism is exhibited by [Pt(NH3)2Cl2]?
Which one of the following pairs represents linkage isomers?
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
What is linkage isomerism? Explain with an example.
The compound(s) that exhibit(s) geometrical isomerism is (are):
(I) [Pt(en)Cl2]
(II) [Pt(en)2]Cl2
(III) [Pt(en)2Cl2]
(IV) [Pt(NH3)2Cl2]
Identify the CORRECT statement about the following complex of platinum.
[PtCl2(en)2]2+
Which of the following does NOT show optical isomerism?
The formula of two complexes X and Y of chromium are given below:
\[\ce{\underset{(X)}{[Cr(H2O)6]Cl3}}\] and \[\ce{\underset{(Y)}{[Cr(H2O)5Cl]Cl2.H2O}}\]
X and Y are examples of ____________ isomers.
Indicate the complex ion which shows geometrical isomerism.
The correct structure of Fe(CO)5 is
Which of the following does not show optical isomerism?
Complex [COCl2(en)2]+ can
Which of the following compound show optical isomerism?
Which of the following has an optical isomer?
Which of the following are isostructural pairs?
(A) \[\ce{SO^{2-}4}\] and \[\ce{CrO^{2-}4}\]
(B) SiCl4 and TiCl4
(C) NH3 and \[\ce{NO^-3}\]
(D) BCl3 and BrCl3
Which compound would exhibit optical isomers?
Write structures for geometrical isomers of diamminebromochloroplatinum (II).
Draw the structure of trans isomers of Pt(NH3)2Cl2.
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Pt(NH3)4Cl2]Br2 and [Pt(NH3)4 Br2]Cl2}\]
Which one of the following complex ions has geometrical isomers?
