Advertisements
Advertisements
प्रश्न
What is linkage isomerism? Explain with an example.
उत्तर
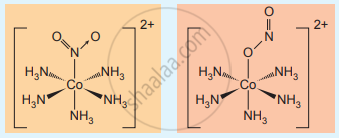
Linkage isomers
Linkage isomerism arises when an ambidentate ligand is bonded to the central metal atom/ion through either of its two different donor atoms.
e.g. - [Co(NH3)5NO2]2+ Pentaammine nitrito-κN Cobalt (III) ion – N attached
[Co(NH3)5ONO2]2+ Pentaammine nitrito-κO Cobalt (III) ion – O attached
APPEARS IN
संबंधित प्रश्न
Draw the geometrical isomers of the following complexes [Pt(NH3)(H2O)Cl2].
Which one of the following will give a pair of enantiomorphs?
Which kind of isomerism is possible for a complex [Co(NH3)4Br2]Cl?
Which would exhibit coordination isomerism?
What is the number of moles of silver chloride precipitated when excess of aqueous silver nitrate is treated with [Co(NH3)4Cl2]Cl?
Indicate the complex ion which shows geometrical isomerism.
Which of the following has an optical isomer?
Which of the following shows maximum number of isomers?
Write structures for geometrical isomers of diamminebromochloroplatinum (II).
Give a chemical test to show that \[\ce{[Co(NH3)5Cl]SO4}\] and \[\ce{[Co(NH3)5SO4]CI}\] are ionisation isomers.
