Advertisements
Advertisements
Question
What is linkage isomerism? Explain with an example.
Solution
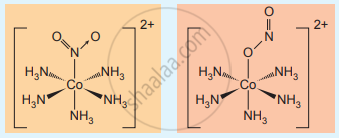
Linkage isomers
Linkage isomerism arises when an ambidentate ligand is bonded to the central metal atom/ion through either of its two different donor atoms.
e.g. - [Co(NH3)5NO2]2+ Pentaammine nitrito-κN Cobalt (III) ion – N attached
[Co(NH3)5ONO2]2+ Pentaammine nitrito-κO Cobalt (III) ion – O attached
APPEARS IN
RELATED QUESTIONS
Give one chemical test as an evidence to show that [Co (NH3)5Cl] are ionisation isomers.
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits ____________ isomerism
Write the type of isomerism exhibited by [Co(NH3)5(NO2)]2+ and [Co(NH3)5ONO]2+ pair of complex ion.
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
The number of geometrical isomers of [CrCl2(en)2]+ is ____________.
Indicate the complex ion which shows geometrical isomerism.
Assertion: Complexes of MX6 and MX5L type (X and L are unidentate) do not show geometrical isomerism.
Reason: Geometrical isomerism is not shown by complexes of coordination number 6.
White precipitate of AgCl dissolves in aqueous ammonia solution due to formation of ______.
Give cis isomer of [Co(NH3)4Cl2]⊕.
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
