Advertisements
Advertisements
Question
Assertion: Complexes of MX6 and MX5L type (X and L are unidentate) do not show geometrical isomerism.
Reason: Geometrical isomerism is not shown by complexes of coordination number 6.
Options
Assertion and reason both are true, reason is correct explanation of assertion.
Assertion and reason both are true but reason is not the correct explanation of assertion.
Assertion is true, reason is false.
Assertion is false, reason is true.
Solution
Assertion and reason both are true but reason is not the correct explanation of assertion.
Explanation:
For complexes of MX6 and MX5L type, different geometric arrangements of the ligands is not possible.
MA4B2, M(AA)2B2 and MA3B3 type of complexes are the complexes with coordination number 6 which show geometrical isomerism.
APPEARS IN
RELATED QUESTIONS
Out of 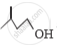 and
and 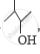 , which one is optically active and why ?
, which one is optically active and why ?
Write the type of isomerism exhibited by [Co(NH3)5(NO2)]2+ and [Co(NH3)5ONO]2+ pair of complex ion.
What are hydrate isomers? Explain with an example.
The term anomers of glucose refer to ____________.
Which of the following is NOT a pair of structural isomers?
Consider the two complexes given below:
\[\ce{\underset{(I)}{[Co(NH3)5SO4]Br}}\] and \[\ce{\underset{(II)}{[Co(NH3)5Br]SO4}}\]
I and II are ____________ isomers.
Which of the following has zero dipole moment?
The relationship between compound (i) and (ii) is
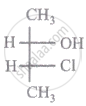 |
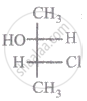 |
| (i) | (ii) |
Write the name of isomerism in the following complexes:
[Cu(NH3)4] [PtCl4] and [Pt(NH3)4] [ CuCl4]
Match the pairs in column I (pairs of isomers) and column II (types of isomers)
| Column I (Pairs of isomers) |
Column II (Types of isomers) |
| (A) [Cr(H2O)5Cl]Cl2.H2O and [Cr(H2O)4Cl2]Cl.2H2O | (i) Ionization isomers |
| (B) [Co(en)2(NO2)2]+ and [Co(en)2(ONO2)]+ | (ii) Hydrate isomers |
| (C) [Co(NH3)6] [Cr(CN)6] and [Cr(NH3)6] [Co(CN)6] | (iii) Linkage isomers |
| (D) [Pt(NH3)4Cl2] Br2 and [Pt(NH3)4Br2]Cl2 | (iv) Coordination isomers |
