Advertisements
Advertisements
Question
Out of 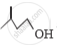 and
and 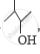 , which one is optically active and why ?
, which one is optically active and why ?
Solution
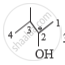 3 - Methy butuan -2- ol is optically active as the 2nd carbon is connected to an hydroxyl
3 - Methy butuan -2- ol is optically active as the 2nd carbon is connected to an hydroxyl
group, methyl group, hydrogen and an alkyl group. It has a chiral carbon.
APPEARS IN
RELATED QUESTIONS
Choose the most correct option.
Which of the following complexes exist as cis and trans isomers?
1. [Cr(NH3)2Cl4]-
2. [Co(NH3)5Br]2⊕
3. [PtCl2Br2]2- (square planar)
4. [FeCl2(NCS)2]2- (tetrahedral)
Which one of the following will give a pair of enantiomorphs?
How many geometrical isomers are possible for \[\ce{[Pt(Py)(NH3)(Br)(Cl)]}\]?
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
Which of the following does NOT show optical isomerism?
Assertion: Complexes of MX6 and MX5L type (X and L are unidentate) do not show geometrical isomerism.
Reason: Geometrical isomerism is not shown by complexes of coordination number 6.
The correct structure of Fe(CO)5 is
The compounds [PtCl2(NH3)4]Br2 and [PtBr2(NH3)4]Cl2 constitutes a pair of ______.
Draw the geometrical isomers of the following complexes [Co(NH3)4Cl2]+
Give a chemical test to show that \[\ce{[Co(NH3)5Cl]SO4}\] and \[\ce{[Co(NH3)5SO4]CI}\] are ionisation isomers.
