Advertisements
Advertisements
Question
Draw all possible geometrical isomers of the complex \[\ce{[Co(en)2Cl2]^+}\] and identify the optically active isomer.
Solution
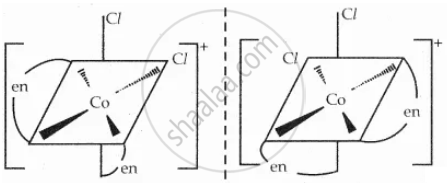
Two Cis \[\ce{[Co(en)2Cl2]^+}\] isomers - optically active
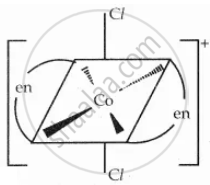
One trans \[\ce{[Co(en)2Cl2]^+}\] isomers - optically inactive
APPEARS IN
RELATED QUESTIONS
Answer in brief.
What are ionization isomers ? Give an example.
Draw isomers of the following.
\[\ce{Pt(NH3)2ClNO2}\]
Answer the following question.
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)2ClBr]2⊕
Define the term Co-ordination isomer.
Which one of the following complexes is not expected to exhibit isomerism?
Fac-mer isomerism is shown by
Identify the CORRECT statement about the following complex of platinum.
[PtCl2(en)2]2+
Which of the following has an optical isomer?
Which of the following shows maximum number of isomers?
Which of the following are isostructural pairs?
(A) \[\ce{SO^{2-}4}\] and \[\ce{CrO^{2-}4}\]
(B) SiCl4 and TiCl4
(C) NH3 and \[\ce{NO^-3}\]
(D) BCl3 and BrCl3
