Advertisements
Advertisements
Question
Answer the following question.
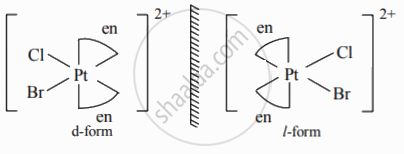
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)2ClBr]2⊕
Solution
Geometric isomers of [Pt(en)2ClBr]2⊕:

Enantiomers of [Pt(en)2ClBr]2⊕:
RELATED QUESTIONS
Write the structural formula and IUPAC names of all possible isomers of the compound with molecular formula C3H8O.
Why dextro and laevo rotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
Answer the following in one or two sentences.
Consider the complexes \[\ce{[Cu(NH3)4][PtCl4] and [Pt(NH3)4] [CuCl4]}\]. What type of isomerism these two complexes exhibit?
Draw isomers of the following.
\[\ce{Pt(NH3)2ClNO2}\]
Answer the following question.
Draw isomers of the following
Ru(NH3)4Cl2
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits ____________ isomerism
Define the term Co-ordination isomer.
Which one of the following will give a pair of enantiomorphs?
Which one of the following pairs represents linkage isomers?
Fac-mer isomerism is shown by
The term anomers of glucose refer to ____________.
Which of the following is NOT an isomer of n-hexane?
____________ isomers are formed when the ligand has two different donor atoms.
The formula of two complexes X and Y of chromium are given below:
\[\ce{\underset{(X)}{[Cr(H2O)6]Cl3}}\] and \[\ce{\underset{(Y)}{[Cr(H2O)5Cl]Cl2.H2O}}\]
X and Y are examples of ____________ isomers.
How many donor groups are present in diethylene triamine?
What type of isomerism is present between (I) [Cr(H2O)6]Cl3 and (II) [Cr(H2O)5Cl]Cl2.H2O?
\[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2 Cl(NO2)]}\] is ______.
Geometrical isomerism is not shown by
The correct structure of Fe(CO)5 is
Which of the following compound show optical isomerism?
Which of the following are isostructural pairs?
(A) \[\ce{SO^{2-}4}\] and \[\ce{CrO^{2-}4}\]
(B) SiCl4 and TiCl4
(C) NH3 and \[\ce{NO^-3}\]
(D) BCl3 and BrCl3
The number of geometrical isomers of \[\ce{[Co(NH3)3 (NO3)3]}\] are ______.
Which among the following solid is a non-polar solid?
Give trans isomer of [Co(NH3)4Cl2]⊕.
Write structures for geometrical isomers of diamminebromochloroplatinum (II).
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Co(NH3)5 [ONO]Cl2 and [Co(NH3)5(NO2)]Cl2}\]
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Cr(H2O)5Cl]Cl2H2O and [Cr(H2O)4Cl2]Cl {.} 2H2O}\]
Which one of the following complex ions has geometrical isomers?
