Advertisements
Advertisements
Question
Give trans isomer of [Co(NH3)4Cl2]⊕.
Solution

trans isomer
APPEARS IN
RELATED QUESTIONS
Out of 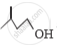 and
and 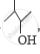 , which one is optically active and why ?
, which one is optically active and why ?
Answer the following in one or two sentences.
Consider the complexes \[\ce{[Cu(NH3)4][PtCl4] and [Pt(NH3)4] [CuCl4]}\]. What type of isomerism these two complexes exhibit?
Answer in brief.
What are ionization isomers ? Give an example.
Answer the following question.
Draw isomers of the following
Ru(NH3)4Cl2
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits ____________ isomerism
Define the term Hydrated isomers.
Draw the geometrical isomers of the following complexes [Pt(NH3)(H2O)Cl2].
Draw optical isomers of [Co(en)3]3+.
How many geometrical isomers are possible for \[\ce{[Pt(Py)(NH3)(Br)(Cl)]}\]?
Fac-mer isomerism is shown by
What is linkage isomerism? Explain with an example.
Explain optical isomerism in coordination compounds with an example.
Which of the following does NOT show optical isomerism?
The complex ions [Co(H2O)5(ONO)]2+ and [Co(H2O)5NO2]2+ are ____________.
Which of the following has zero dipole moment?
Indicate the complex ion which shows geometrical isomerism.
\[\ce{IUPAC}\] name of \[\ce{[Pt(NH3)2 Cl(NO2)]}\] is ______.
Complex [COCl2(en)2]+ can
Which of the following has an optical isomer?
Which of the following shows maximum number of isomers?
\[\ce{CH3CH2COO- Na+ ->[NaOH, + ?][Heat] CH3CH3 + Na2CO3}\]
Consider the above reaction and identify the missing reagent/chemical.
The one that is not expected to show isomerism is ______.
The compounds [PtCl2(NH3)4]Br2 and [PtBr2(NH3)4]Cl2 constitutes a pair of ______.
Write structures for geometrical isomers of diamminebromochloroplatinum (II).
Explain the geometrical isomerism of the octahedral complex of the type [M(AA)2B2]n± with a suitable example.
What are structural isomers or constitutional isomers?
Draw the structure of cis isomers of Pt(NH3)2Cl2.
Draw the structure of trans isomers of Pt(NH3)2Cl2.
