Advertisements
Advertisements
प्रश्न
Give trans isomer of [Co(NH3)4Cl2]⊕.
उत्तर

trans isomer
APPEARS IN
संबंधित प्रश्न
Out of 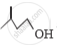 and
and 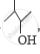 , which one is optically active and why ?
, which one is optically active and why ?
Give one chemical test as an evidence to show that [Co (NH3)5Cl] are ionisation isomers.
Why dextro and laevo rotatory isomers of Butan-2-ol are difficult to separate by fractional distillation?
Answer the following in one or two sentences.
Predict whether the [Cr(en)2(H2O)2]3+ complex is chiral. Write the structure of its enantiomer.
Answer the following in one or two sentences.
Consider the complexes \[\ce{[Cu(NH3)4][PtCl4] and [Pt(NH3)4] [CuCl4]}\]. What type of isomerism these two complexes exhibit?
Answer in brief.
What are ionization isomers ? Give an example.
Draw isomers of the following.
\[\ce{Pt(NH3)2ClNO2}\]
Write the type of isomerism exhibited by [Co(NH3)5(NO2)]2+ and [Co(NH3)5ONO]2+ pair of complex ion.
Which type of isomerism is exhibited by [Pt(NH3)2Cl2]?
Which kind of isomerism is possible for a complex [Co(NH3)4Br2]Cl?
The term anomers of glucose refer to ____________.
Which would exhibit coordination isomerism?
How many isomers are possible for an alkane having molecular formula C5H12?
How many donor groups are present in diethylene triamine?
What type of isomerism is present between (I) [Cr(H2O)6]Cl3 and (II) [Cr(H2O)5Cl]Cl2.H2O?
The relationship between compound (i) and (ii) is
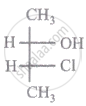 |
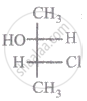 |
| (i) | (ii) |
Which of the following compound show optical isomerism?
Which of the following has an optical isomer?
Which of the following shows maximum number of isomers?
Which of the following compound is optically active?
Which compound would exhibit optical isomers?
Define Distereoisomers.
Match the pairs in column I (pairs of isomers) and column II (types of isomers)
| Column I (Pairs of isomers) |
Column II (Types of isomers) |
| (A) [Cr(H2O)5Cl]Cl2.H2O and [Cr(H2O)4Cl2]Cl.2H2O | (i) Ionization isomers |
| (B) [Co(en)2(NO2)2]+ and [Co(en)2(ONO2)]+ | (ii) Hydrate isomers |
| (C) [Co(NH3)6] [Cr(CN)6] and [Cr(NH3)6] [Co(CN)6] | (iii) Linkage isomers |
| (D) [Pt(NH3)4Cl2] Br2 and [Pt(NH3)4Br2]Cl2 | (iv) Coordination isomers |
The co-ordination number of Co3+ ion in the complex [Co(NH3)4Cl2]⊕ is ______.
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Cr(H2O)5Cl]Cl2H2O and [Cr(H2O)4Cl2]Cl {.} 2H2O}\]
Which one of the following complex ions has geometrical isomers?
