HSC Science (General)
HSC Science (Electronics)
HSC Science (Computer Science)
Academic Year: 2022-2023
Date & Time: 1st March 2023, 11:00 am
Duration: 3h
Advertisements
General instructions :
The question paper is divided into four sections :
- Section A: Q. No. 1 contains Ten multiple-choice type of questions carrying One mark each. Q. No. 2 contains Eight very short answer type of questions carrying One mark each.
- Section B: Q. No. 3 to Q. No. 14 contain Twelve short answer type of questions carrying Two marks each. (Attempt any Eight).
- Section C: Q. No. 15 to Q. No. 26 contains Twelve short answer type of questions carrying Three marks each. (Attempt any Eight).
- Section D: Q. No. 27 to Q. No. 31 contain Five long answer type of questions carrying Four marks each. (Attempt any Three).
- Use of the log table is allowed. Use of calculator is not allowed.
- Figures to the right indicate full marks.
- For each multiple-choice type of question, it is mandatory to write the correct answer along with its alphabet. e.g., (a)......../(b)......../(c)......./(d)........
No marks(s) shall be given if ONLY the correct answer or the alphabet of the correct answer is written. Only the first attempt will be considered for evaluation.
Given:
R = 8.314 J.K–1. mol–1
NA = 6.022 × 1023
F = 96500C
The relation between the radius of the sphere and the edge length in the body-centred cubic lattice is given by the formula ______.
`sqrt(3)r` = 4a
r = `sqrt(3)/a xx 4`
r = `sqrt(3)/4a`
r = `sqrt(2)/4 xx a`
Chapter: [0.01] Solid State
The pH of a weak monoacidic base is 11.2, and its OH– ion concentration is ______.
1.585 × 10–3 mol dm–3
3.010 × 10–11 mol dm–3
3.010 × 10–3 mol dm–3
1.585 × 10–11 mol dm–3
Chapter: [0.03] Ionic Equilibria
Which of the following correctly represents integrated rate law equation for a first order reaction in a gas phase?
k = `2.303/t xx log_10 P_i/(P_i - P)`
k = `2.303/t xx log_10 P_i/(2P_i - P)`
k = `2.303/t xx log_10 (2P_i)/(P_i - P)`
k = `2.303/t xx log_10 (P_i - P)/(2P_i)`
Chapter: [0.06] Chemical Kinetics
The spin only magnetic moment of Mn2+ ion is ______.
4.901 BM
5.916 BM
3.873 BM
2.846 BM
Chapter: [0.08] Transition and Inner Transition Elements
The correct formula of a complex having IUPAC name Tetraamminedibromoplatinum (IV) bromide is ______.
[Pt Br (NH3)4] Br2
[Pt Br2 (NH3)4] Br
[Pt Br2 (NH3)4] Br2
[Pt Br (NH3)4] Br
Chapter: [0.09] Coordination Compounds
The allylic halide, among the following, is ______.
\[\begin{array}{cc}
\phantom{}\ce{R - CH - R}\phantom{}\\
|\phantom{..}\\
\ce{X}\phantom{..}
\end{array}\]
CH2 = CH – X
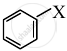
CH2 = CH – CH2 – X
Chapter: [0.1] Halogen Derivatives
The product of following reaction is
\[\ce{CH3 - CH = CH - CH2 - CHO ->[i) LiAlH4][ii) H3O+]}\] ______?
CH3 – CH2 – CH2 – CH2 – CH2 – OH
CH3 – CH = CH – CH2 – CH2 – OH
CH3 – CH2 – CH2 – CH2 – COOH
CH3 – CH = CH – CH2 – COOH
Chapter: [12.01] Aldehydes and Ketones
Ozonolysis of 2, 3 dimethyl but-2-ene, followed by decomposition by Zn dust and water gives ______.
acetaldehyde
propionaldehyde and acetone
acetone
acetaldehyde and butyraldehyde
Chapter: [0.12] Aldehydes, Ketones and Carboxylic Acids [12.01] Aldehydes and Ketones
The glycosidic linkage present in maltose is ______.
α, β-1, 2-glycosidic linkage
α-1, 4-glycosidic linkage
β-1, 4-glycosidic linkage
α-1, 6-glycosidic linkage
Chapter: [0.14] Biomolecules
The monomer of natural rubber is ______.
Isoprene
Acrylonitrile
ε-Caprolactam
Tetrafluoroethylene
Chapter: [0.15] Introduction to Polymer Chemistry [0.15] Polymers
Write the name of the technique used to know geometry of nanoparticles.
Chapter: [0.16] Green Chemistry and Nanochemistry
Write the name of the product formed by the action of LiAlH4/ether on acetamide.
Chapter: [0.13] Amines [13.01] Amines
Write the structure of the product formed when chlorobenzene is treated with sodium metal in the presence of dry ether.
Chapter: [0.1] Halogen Derivatives
Write the chemical composition of cryolite.
Chapter: [0.07] Elements of Groups 16, 17 and 18
Write the name of platinum complex used in the treatment of cancer.
Chapter: [0.09] Coordination Compounds
Write the SI unit of the cryoscopic constant.
Chapter: [0.02] Solutions
Write the correct condition for spontaneity in terms of Gibbs energy.
Chapter: [0.04] Chemical Thermodynamics
Calculate molar conductivity for 0.5 M BaCl2 if its conductivity at 298K is 0.01 Ω–1 cm–1.
Chapter: [0.05] Electrochemistry
Distinguish between lanthanoid and actinoids.
Chapter: [0.08] Transition and Inner Transition Elements [8.02] F-block Elements
Advertisements
Calculate the mole fraction of solute, if the vapour pressure of pure benzene at a certain temperature is 640 mmHg and the vapour pressure of a solution of a solute in benzene is 600 mmHg.
Chapter: [0.02] Solutions
Define Green chemistry.
Chapter: [0.16] Green Chemistry and Nanochemistry
Write any three advantages of nanoparticles and nanotechnology.
Chapter: [0.16] Green Chemistry and Nanochemistry
Explain the following term:
Substitutional impurity defect
Chapter: [0.01] Solid State
Explain the following term:
Interstitial impurity defect
Chapter: [0.01] Solid State
Write the chemical reaction for the following:
Chlorobenzene is heated with fuming H2SO4.
Chapter: [0.1] Halogen Derivatives
What is the action of the following on ethyl bromide:
silver acetate
Chapter: [10.01] Haloalkanes
Define Acidic buffer solution.
Chapter: [0.03] Ionic Equilibria
Write the relationship between solubility and solubility product for PbI2.
Chapter: [0.03] Ionic Equilibria
What is the action of the following reagents on ethyl amine?
Chloroform and caustic potash
Chapter: [13.01] Amines
What is the action of the following reagents on ethyl amine?
Nitrous acid
Chapter: [13.01] Amines
Calculate standard Gibbs energy change at 25°C for the cell reaction.
\[\ce{Cd_{(s)} + Sn^{2+}_{ (aq)} -> Cd^{2+}_{ (aq)} + Sn_{(s)}}\]
\[\ce{E^0_{cd}}\] = –0.403 V, \[\ce{E^0_{Sn}}\] = –0.136 V
Chapter: [0.05] Electrochemistry
Explain the preparation of glucose from sucrose.
Chapter: [0.14] Biomolecules
Write the structure of D-ribose.
Chapter: [0.14] Biomolecules
Define the Extensive properties.
Chapter: [0.04] Chemical Thermodynamics
Calculate the work done during the expansion of 2 moles of an ideal gas from 10 dm3 to 20 dm3 at 298 K in a vacuum.
Chapter: [0.04] Chemical Thermodynamics
Write the preparation of nylon 6, 6.
Chapter: [0.15] Introduction to Polymer Chemistry
Draw the structure of chloric acid.
Chapter: [0.07] Elements of Groups 16, 17 and 18
Draw the structure of the following compound:
Peroxy disulphuric acid
Chapter: [0.07] Elements of Groups 16, 17 and 18
Explain the term osmosis.
Chapter: [0.02] Solutions [0.02] Solutions and Colligative Properties
How will you determine the molar mass of non-volatile solute by elevation of boiling point?
Chapter: [0.02] Solutions
Advertisements
Convert the following:
Ethyl alcohol into ethyl acetate
Chapter: [0.11] Alcohols, Phenols and Ethers [11.01] Alcohols [11.02] Phenols [11.03] Ethers
How is phenol converted into the following?
benzene
Chapter: [0.11] Alcohols, Phenols and Ethers [11.01] Alcohols [11.02] Phenols [11.03] Ethers
How will you obtain the following? (Give balanced equation.)
Ethyl chloride from diethyl ether.
Chapter: [11.03] Ethers
A weak monobasic acid is 10% dissociated in 0.05 M solution. What is the percent dissociation in 0.15 M solution?
Chapter: [0.03] Ionic Equilibria
Explain the dehydrohalogenation reaction of 2-chlorobutane.
Chapter: [0.1] Halogen Derivatives
Write environmental effect of CFC.
Chapter: [0.1] Halogen Derivatives
2000 mmol of an ideal gas expanded isothermally and reversibly from 20 L to 30 L at 300 K, calculate the work done in the process (R = 8.314 JK–1 mol–1).
Chapter: [0.04] Chemical Thermodynamics
What are interstitial compounds?
Chapter: [0.08] Transition and Inner Transition Elements
Give the classification of alloys with examples.
Chapter: [0.08] Transition and Inner Transition Elements
Draw labelled diagram of H2 – O2 fuel cell.
Chapter: [0.05] Electrochemistry
Write two applications of fuel cells.
Chapter: [0.05] Electrochemistry
Explain the formation of [CoF6]3Θ complex with respect to
- Hybridisation
- Magnetic properties
- Inner/outer complex
- Geometry
Chapter: [0.09] Coordination Compounds
Explain pseudo-first-order reaction with an example.
Chapter: [0.06] Chemical Kinetics
Derive integrated rate law for a zero-order reaction \[\ce{A -> Product}\].
Chapter: [0.06] Chemical Kinetics
Explain Aldol condensation of ethanal.
Chapter: [0.12] Aldehydes, Ketones and Carboxylic Acids
Explain anomalous behaviour of oxygen in group 16 with respect to:
- Atomicity
- Magnetic property
- Oxidation state
Chapter: [0.07] Elements of Groups 16, 17 and 18 [7.02] Group 16 Elements
How will you prepare acetic anhydride from acetic acid?
Chapter: [0.12] Aldehydes, Ketones and Carboxylic Acids
Write chemical reactions for the following conversion:
Acetic acid into ethyl alcohol
Chapter: [0.11] Alcohols, Phenols and Ethers [11.01] Alcohols [11.02] Phenols [11.03] Ethers
Write the IUPAC name of methylphenylamine.
Chapter: [0.13] Amines
Write the structure of methylphenylamine.
Chapter: [0.13] Amines
Show that the time required for 99.9% completion of a first-order reaction is three times the time required for 90% completion.
Chapter: [0.06] Chemical Kinetics
Give the electronic configuration of gadolinium (Z = 64).
Chapter: [0.08] Transition and Inner Transition Elements
Answer the following
Which nanomaterial is used for tyres of the car to increase the life of tyres?
Chapter: [0.16] Green Chemistry and Nanochemistry
Obtain the relationship between ΔH and ΔU for gas phase reactions.
Chapter: [0.04] Chemical Thermodynamics
Answer the following in one sentence.
Define the term: Vulcanization
Chapter: [0.15] Introduction to Polymer Chemistry [0.15] Polymers
Silver crystallizes in the fcc structure. If the edge length of the unit cell is 400 pm, calculate the density of silver (Atomic mass of Ag = 108).
Chapter: [0.01] Solid State
Explain haloform reaction with suitable example.
Chapter: [0.12] Aldehydes, Ketones and Carboxylic Acids
Define Distereoisomers.
Chapter: [0.09] Coordination Compounds
Give cis isomer of [Co(NH3)4Cl2]⊕.
Chapter: [0.09] Coordination Compounds
Give trans isomer of [Co(NH3)4Cl2]⊕.
Chapter: [0.09] Coordination Compounds
Define Reference electrode
Chapter: [0.05] Electrochemistry
Give reason:
Bleaching action of ozone is also called dry bleach.
Chapter: [0.07] Elements of Groups 16, 17 and 18
Write Dow process for the preparation of Phenol.
Chapter: [11.02] Phenols
What is the action of bromine water on carbolic acid?
Chapter: [11.02] Phenols
Elements of group 16 have lower ionization enthalpy values compared to those of group 15 elements. Explain why?
Chapter: [0.07] Elements of Groups 16, 17 and 18
Write two uses of dioxygen.
Chapter: [7.02] Group 16 Elements
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
Maharashtra State Board previous year question papers 12th Standard Board Exam Chemistry with solutions 2022 - 2023
Previous year Question paper for Maharashtra State Board 12th Standard Board Exam Chemistry-2023 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of Maharashtra State Board 12th Standard Board Exam.
How Maharashtra State Board 12th Standard Board Exam Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
