Advertisements
Advertisements
प्रश्न
Convert the following:
Ethyl alcohol into ethyl acetate
उत्तर
Ethyl alcohol reacts with acetic acid to give ethyl acetate.
\[\ce{\underset{Ethyl alcohol}{C2H5OH} + \underset{Acetic acid or Ethanoic acid}{CH3COOH} <=>[H^+] \underset{ethyl acetate}{CH3COOC2H5} + H2O}\]
APPEARS IN
संबंधित प्रश्न
What is metamerism?
Explain metamerism with suitable examples of ethers
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\phantom{............}\ce{CH2OH}\\
\phantom{......}|\\
\ce{CH3 - CH2 - CH - CH - CH - CH3}\\
\phantom{......}|\phantom{............}|\phantom{.}\\
\phantom{........}\ce{CH2Cl}\phantom{......}\ce{CH3}\phantom{}
\end{array}\]
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\phantom{........................}\ce{CH2OH}\\
\phantom{..................}|\\
\ce{CH3 - CH - CH2 - CH - CH - CH3}\\
\phantom{}|\phantom{.............}|\phantom{........}\\
\phantom{..}\ce{CH3}\phantom{..........}\ce{OH}\phantom{........}
\end{array}\]
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\ce{H2C = CH - CH - CH2 - CH2 - CH3}\\
|\phantom{..........}\\
\ce{OH}\phantom{........}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{.}}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
|\phantom{......}|\phantom{..}\\
\ce{OH}\phantom{...}\ce{OH}\phantom{}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{..}\\
\ce{OH}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - O - CH2 - CH - CH3}\\
\phantom{..........}|\\
\phantom{............}\ce{CH3}
\end{array}\]
Write IUPAC name of the following compound:
C6H5 – O – C2H5
Write IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
Give IUPAC name of the following ether:
\[\begin{array}{cc}
\ce{C2H5OCH2 - CH - CH3}\\
\phantom{.....}|\\
\phantom{.......}\ce{CH3}
\end{array}\]
Give IUPAC name of the following ether:
CH3OCH2CH2Cl
Give IUPAC name of the following ether:
CH3CH2CH2OCH3
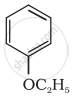
Give IUPAC name of the following ether:
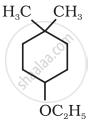
Give IUPAC name of the following ether:
Ethylidene dichloride when boiled with aqueous solution of NaOH yields _______.
(A) formaldehyde
(B) acetaldehyde
(C) acetone
(D) ethyl methyl ketone
Natalite is a mixture of
(a) diethyl ether and methanol
(b) diethyl ether and ethanol
(c) dimethyl ether and methanol
(d) dimethyl ether and ethanol
Write the structure and IUPAC name of 'methyl-n-propyl ether'.
What is the action of hot HI on it?
3-Methylbutane-2-ol on heating with HI gives ______
How is phenol converted into the following?
benzene
How is phenol converted into the following?
benzoquinone
How is phenol converted into the following?
picric acid
Write the IUPAC name of the following :
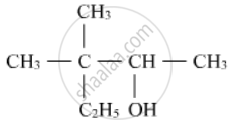
How do you convert the Ethanal to Propanone
In the dehydration of alcohols to alkenes by heating with concentrated sulphuric acid, the initiation step is:
(1) formation of carbonation
(2) formation of an ester
(3) protonation of the alcohol molecule
(4) elimination of water
What.will be the product fonned when chlorobenzene is heated with sodium metal in the presence of dry ether?
Propanoic acid to ethylamine.
Resorcinol on distillation with zinc dust gives _________.
Write structural formulae for Methyl vinyl ether.
Write structural formulae for Pentane-1,4-diol
Write structural formulae for Cyclohex-2-en-1-ol.
Write IUPAC name of the following
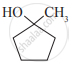
Write IUPAC name of the following
\[\begin{array}{cc}\ce{CH3-CH-CH-CH2-OH}\\|\phantom{.....}|\phantom{.......}\\\ce{OH}\phantom{..}\ce{CH3}\phantom{.....}\end{array}\]
Write IUPAC names of the following
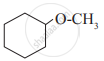
In a carbinol system of nomenclature tert.butyl alcohol is named as _______________
Glycerol is ____________.
Give IUPAC names of the following compound:
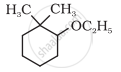
Give IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{...}\ce{CH3}\phantom{.}\ce{H}\phantom{..}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{.}\ce{H}\phantom{...}\ce{OH}\phantom{.}\ce{H}\phantom{.}\\
\end{array}\]
3-methylphenol is called ____________.
One of the following is not a dihydroxy derivative of benzene.
The compound HOCH2 – CH2OH is __________.
An example of a compound with functional group – O – is ____________.
Butane-2-ol is ____________.
Which of the following compounds is oxidised to prepare methyl ethyl ketone?
Ethylene reacts with Baeyer’s reagent to give ______.
When ethyl alcohol reacts with acetic acid, the products formed are:
Which of the following is most acidic?
The IUPAC name of the ether CH2 = CH–CH2OCH3 is:
The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
The product of acid catalysed hydration of 2-phenylpropene is:
The heating of phenyl methyl ether with HI produces:
IUPAC name of the compound is:
\[\begin{array}{cc}
\ce{CH3-CH-OCH3}\\
|\phantom{....}\\
\ce{CH3}\phantom{..}
\end{array}\]
The correct acidic strength order of the following is:
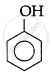
(I)

(II)
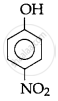
(III)
IUPAC name of m-cresol is ______.
Which of the following compounds will react with sodium hydroxide solution in water?
Arrange the following compounds in decreasing order of acidity.
\[\ce{H2O, ROH, HC ≡ CH}\]
Write steps to carry out the conversion of phenol to aspirin.
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
How can phenol be converted to aspirin?
Write the IUPAC name of the following compound.
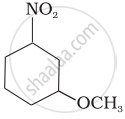
Identify A and B in the following:

How are the following conversions carried out?
Methyl magnesium bromide→2-Methylpropan-2-ol.
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Draw structure of the following compound.
2. 5-Diethylphenol
Draw structure of the following compound.
2-Methoxypropane
Draw structure of the following compound.
Prop-2-en-1-ol
Give the structures of Thiosulphuric acid and Peroxy monosulphuric acid.
Write structural formulae for:
Salicylic acid
Write the IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{.....}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{.}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
